jarun011
Introduction
Results from three major testosterone related studies are due in 2023. They include cardiovascular (CV) safety and efficacy results in Testosterone Replacement Therapy (TRT) for both men and women. I expect an NDA submission for the first approved testosterone drug designed for women to treat Hypoactive Sexual Desire Disorder (HSDD), and possibly other indications. I also expect supplemental NDA submissions for Androgel 1.62% for men. Finally, the findings of a 700,000-participant study assessing the impact of genetically decreasing testosterone levels on dementia and other mental health conditions, such as anxiety and depression is expected.
The findings should help guide physicians in prescribing testosterone, lead to expanded use of testosterone for multiple indications and identify areas warranting further study for expanded treatment. The release of this much testosterone related information should generate interest in testosterone related stocks, driving up PPS, especially for AbbVie (NYSE:ABBV) and ANI Pharmaceuticals (NASDAQ:ANIP).
Testosterone for Men
March 3, 2015, the FDA issued a Drug Safety Communication, cautioning about using testosterone products for low testosterone due to aging, which required a labeling change to inform of possible increased risk of heart attack and stroke. The FDA also required manufacturers of approved testosterone products to conduct clinical trial to more clearly address the question of whether there was an increased risk of heart attack or stroke.
In response, AbbVie led a Testosterone Replacement Therapy (TRT) Manufacturer Consortium including Allergan (now AbbVie) Acerus Pharmaceuticals Corporation (OTCQB:ASPCF) /Aytu BioPharma, Inc. (AYTU); Endo International (OTCPK:ENDPQ); Upsher-Smith Laboratories LLC, in a 5,246-participant study commonly referred to often as the “TRAVERSE” study. The study involving AbbVie’s Androgel 1.62% was completed on November 29, 2022, and may lead to expanded indications for the drug. AbbVie is listed as the sponsor and study director, whereas the other members of the consortium are listed as collaborators. It is unknown what level of collaboration these companies had in the trial.
The primary outcome measurement is listed as time to major adverse cardiac event. The secondary outcome measurement is assessing CV safety. In addition, the trial would assess prostate safety by assessing the incidence of high-grade prostate cancer.
This trial was also assessing efficacy for the following:
- Improvement in sexual activity in hypogonadal men with low libido.
- The remission of depression in hypogonadal men with late-onset, low grade persistent depressive disorder (dysthymia).
- The reduction in incidence of clinical fractures.
- The proportion of anemic men whose anemia is corrected during the intervention period.
- The reduction in progression from pre-diabetes to diabetes in subset of participants with pre-diabetes at baseline.
The Consent to Participate in a Research Study form used to recruit participants, showed that in order for a participant to complete the entire study, it required a 62 month commitment. However, the original start date for the trial was May 3, 2018, and had an estimated completion date of June 2022, a 42-month period. The earliest participants would be at least eighteen months or more shy of the 60-month study timeline. The study originally anticipated enrolling 6,000 participants, however, only 5,246 participants were actually enrolled, with the trial still recruiting as late as February 14, 2022.
AbbVie’s Humira faces biosimilar competition in the US, as early as the end of January 2023. As of October 28, 2022, AbbVie was still projecting a revenue reduction ranging between 35% and 55% for Humira. It appears completion of the trial prior to 2023 was a priority, over obtaining 60 months of safety data, from the beginning.
Testosterone for women
ANIP or an undisclosed partner is expected to release the results of the 3,565 participant study titled Safety and Efficacy of LibiGel® for Treatment of HSDD in Postmenopausal Women (BLOOM). The most recent trial update supports the likelihood that an NDA has been, or will be, submitted with the expectation of becoming the first FDA approved testosterone drug to treat post-menopausal women for HSDD. Currently, no FDA approved testosterone drug exists for women. Nor is there an FDA approve drug to treat post-menopausal women diagnosed with HSDD.
On October 27, 2021, a Certification or Extension request was submitted to delay the submission of results for the trial, which involved post-menopausal women, who had HSDD and were at an elevated risk of experiencing CV events. Though it does not identify whether a Certification or an Extension request was submitted, an extension request is commonly issued when the posting results could impact ongoing trials. There are no ongoing trials. The completion date of the trial was December 2012, so there is no obligation to post results, much less, seek an extension to delay posting results. Therefore, it is most likely Certification request, which is a precursor to an NDA submission.
A unblinded review with 4,000 patient years of data, showed that TRT in post-menopausal women, who were at risk of experiencing CV events, reduced the number of CV events by at least 70% over expectations. It also reduced the number of CV events by an undisclosed amount over placebo. In 2015, they discovered that TRT reduced breast cancer events by an undisclosed amount over expectations. A finding not evident during the earlier review or when the trial was completed (December 2012). The discoveries are only public in patent applications, listing the Study Director, Dr. Michael C. Snabes (Senior Medical Director with AbbVie Global Clinical Research and Development) as one of the inventors.
Placebo effect sunk the two efficacy trials. However, according to an American Heart Journal article entitled A cardiovascular safety study of LibiGel (testosterone gel) in postmenopausal women with elevated cardiovascular risk and hypoactive sexual desire disorder, the larger study was evaluating efficacy, using questions like those used in the Intrinsa studies. An FDA advisory panel had determined that Intrinsa had established efficacy, however required more safety data to be approved. The LibiGel trial had a surgically menopausal patient population size consistent with the two efficacy trials. Whether the LibiGel data is sufficient to overcome the placebo effect remains to be seen.
In the 18 years since the FDA advisory panel rejected Intrinsa, understanding HSDD has grown significantly. In April 2018, the Mayo Clinic Proceedings published “The International Society for the Study of Women’s Sexual Health (ISSWSH) Process of Care for Management of Hypoactive Sexual Desire Disorder in Women, evidence-based guidelines for diagnosis and treatment of HSDD in women by health care professionals.
Process of Care for Management of HSDD (Mayo Clinic Proceedings)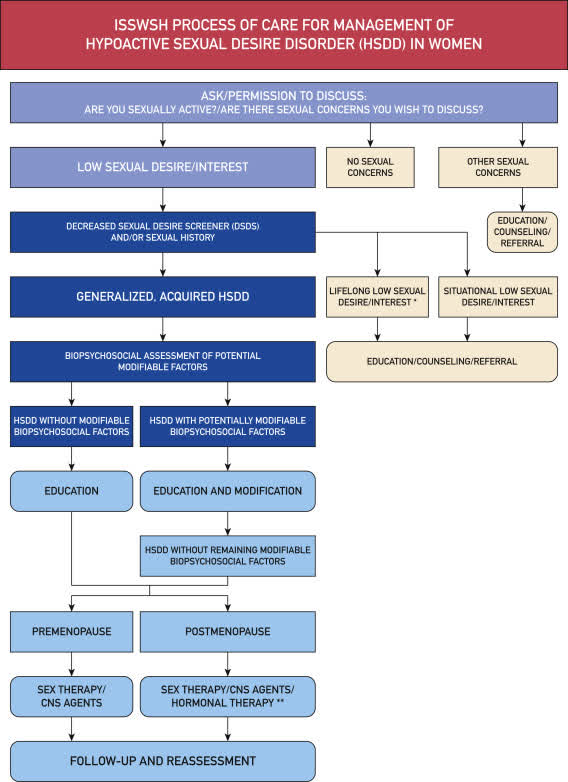
In September 2019, the first Global Consensus Position Statement on the Use of Testosterone Therapy for Women (Position Statement) was released. It provides clinical guidance regarding the use of TRT in women, examining the effect on sexual function, well-being, mood, and cognition, musculoskeletal effects, CV and breast health, as well side effects and adverse events. The Position Statement found that though no established indications for testosterone therapy for women existed, clinicians have been treating women with testosterone for decades, uncertain of the benefits and the risks. In most countries, including the United States, testosterone therapy that is prescribed is off-label male testosterone or compounded therapies, which is not recommenced. Only transdermal or cream applications were recommended. They also found that further investigation is warranted with respect to an individual’s well-being, musculoskeletal and cognitive health, and long-term safety. The LibiGel trial results should resolve the long-term safety questions.
Testosterone Levels, Dementia, and Adverse Mental Health Outcomes
The M.D. Anderson Cancer Center is expected to release the results of a 700,000 participant retrospective study titled Evaluation of Association Between Testosterone Levels, Dementia, and Adverse Mental Health Outcomes, with an estimated completion date of November 1, 2022. This study will evaluate the association between decreasing testosterone levels and risk of dementia and adverse mental health outcomes (e.g., depression and anxiety). They intend to develop a risk assessment tool linking testosterone levels to the studied conditions. The results should assist in developing future trials involving TRT and Androgen Deprivation Therapy.
HSDD Market Opportunity
“Androgen Therapy in Women“, indicated that in 2009, over 4 million prescriptions were written for off label female testosterone drugs or compounded testosterone. In 2011, BioSante Pharmaceuticals believed the market to be more than $2 billion. Estimates were influenced by a survey of doctors that indicated 96% of the prescriptions would be switched to an FDA approved drug and projected growth of the menopausal demographic. “Women’s hormone drug prices have climbed for years, but controversy hasn’t ensued. Why?”, indicates that between 2014 and 2018 estrogen drug prices rose approximately 100%. Prices for an FDA approved female testosterone drug would have kept pace. The $2 billion market has, arguably, grown to a $4 billion market, in the US alone.
Rising Estrogen Drug Prices (U.S. Pharmacist)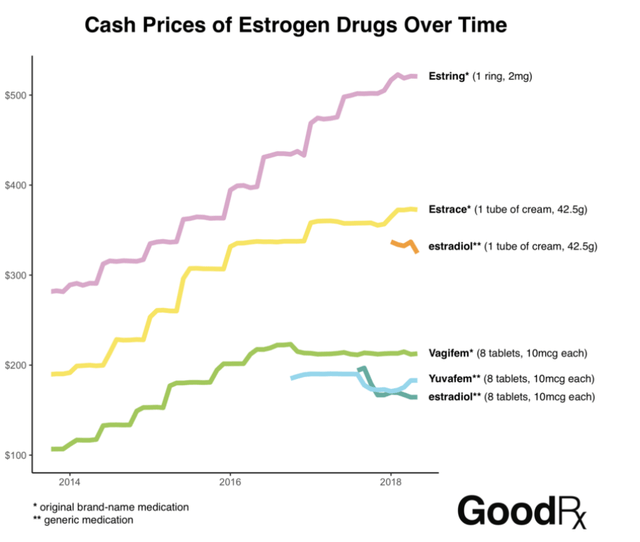
LibiGel Revenue Opportunity (BioSante Pharmaceuticals)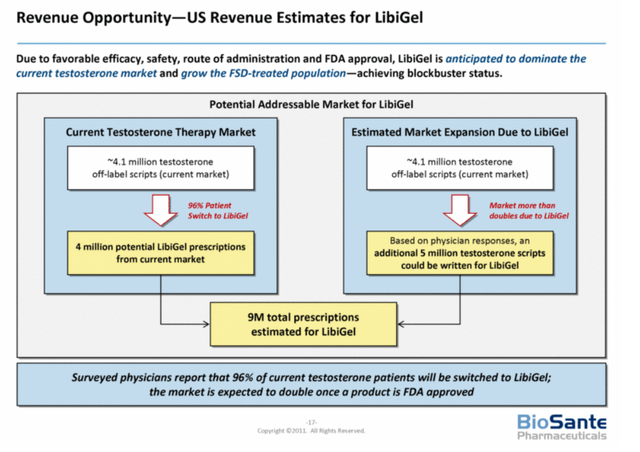
In research conducted by Campbell Alliance in 2011, physicians reported a high need for an effective, safe and FDA approved product to treat HSDD. This market has wide potential with predictions ranging from about $2.0 billion up to $5 billion. Some analyst, had a less optimistic view on the market potential, forecasting sales ranging between $286 million (Leerink Swann) and $841 million (Roth Capital Markets). A $572 million to $1.682 billion market, when keeping pace with the rise in estrogen prices.
Who will take the lead?
In addition to the Certification/Extension request to delay submitting results, small grammatical changes were made to the clinical trial, however, the sponsor is still BioSante Pharmaceuticals. If ANIP were advancing it, they would have identified themselves as the sponsor. ANIP would also need a large pharmaceutical partner to address the global demand.
AbbVie is a possibility, as Dr. Snabes has worked for AbbVie since he left BioSante Pharmaceuticals in 2013. AbbVie now owns the Intrinsa portfolio (through Allergan). Intrinsa (patch), like LibiGel (gel) involved administering 300 mcg of testosterone daily. Intrinsa’s clinical trial evidence is the second largest body of clinical data in relation to female testosterone, behind LibiGel. The combined information would only strengthen an NDA submission.
However, ANIP has also established a good working relationship with Merck & Co. (MRK) after acquiring the discontinued Purified Cortrophin Gel in 2016 and have worked with Merck to get it reapproved in November 2021. Following the approval, in February 2022, after 24 years with Merck, Elizabeth Powell left her position as Managing Counsel/Executive Direction for US Commercial and Compliance Legal with Merck to work for ANIP as their Chief Compliance Officer and Head of Legal for Rare Diseases. A partnership with Merck would be well served by hiring Powell. Merck’s website list Oncology and Cardio-metabolic disorders as areas of focus. The discovery that TRT reduces CV and Breast Cancer risk, and events would tuck in nicely to Merck’s pipeline. Incidentally, Merck still lists Women’s Health as a Key Therapeutic Area on the Pipeline webpage with no product listed even after spinning off Organon in May 2021.
Could ANIP have a deal with AbbVie and Merck?
Issues
Androgel 1.62% has generic competition. Therefore, it can be argued that AbbVie conducted the Traverse study solely to meet FDA requirements and was completed early to preserve cash. Which is a possibility, but the efficacy results may prove too good to walk away from and the safety data should differentiate it from its competitors.
Shortly after the Position Statement was released, companies who stood the most to gain, inexplicably let their patents expire by not paying their maintenance fees. Antares Pharmaceuticals (now Halozyme) allowed their patent titled Methods and Apparatus for Transdermal or Transmucosal Application of Testosterone to expire for non-payment of the maintenance fee of $1,504 on 2020-01-06. The patent claims to provide a method for treating hypoactive sexual desire disorder (HSDD) or female sexual dysfunction (FSD) in a female in need thereof by daily administration testosterone. This patent has an adjusted expiration date of 2028-12-30 which likely would have been extended another 5 years through the FDA Patent Restoration Program.
Noven Pharmaceuticals’ transdermal patch technology was used for Intrinsa. Oddly, on April 26, 2020, a little over seven months after the Position Statement was issued, Noven Pharmaceuticals’ patent titled “Transdermal Testosterone Device and Delivery” was allowed to expire for non-payment of the $2,000 maintenance fee. This patent was for a 50% smaller version of the Intrinsa patch, previously marketed by Warner Chilcott (now AbbVie) in Europe. Warner Chilcott conducted a clinical trial titled Bioequivalence of Testosterone Reduced-size Patch Relative to the Testosterone Reference Patch in support of the patent application. The patent was set to expire March 14, 2032. It too could possibly have been extended another 5 years up to March 14, 2037.
The timing of letting the patents expire, especially when considering how little the maintenance fees cost, leads me to believe the patents have been acquired and will soon be reinstated.
Conclusion
I believe testosterone replacement will be a big story in 2023. The large CV related trial results for both men and women should draw a lot of attention and generate investor interest in related stocks. AbbVie is an interesting play, as Humira’s revenue loss is factored into the current share. However, if the results show efficacy in any or all in reducing depression, clinical fractures, and progression from pre-diabetes to diabetes, in addition to correcting anemia levels, concerns about Humira revenue erosion become less concerning. AbbVie also appears best positioned to take the lead in bringing the first female testosterone drug to market. If this is in AbbVie’s future, AbbVie’s share should climb much higher, especially with positive CV and Breast cancer reduction announcements and no generic competition. ANIP should also see a significant influx of investors once the results are released. With only 17.4 million outstanding shares, PPS should rise rapidly.
Though not the focus of this article, the other member of the consortium may also benefit, eventually. The M.D. Anderson Cancer Center study is remarkably interesting, as it will likely put added pressure on the FDA to approve a female testosterone so that cost-effective real-world studies can be conducted for men and women. Which no doubt AbbVie is more than capable of taking the lead. The three studies should lead to increased demand for testosterone testing, which should benefit suppliers and manufacturers.
JPMorgan’s Healthcare Conference runs from January 9 to 12, 2023. It is the only presentation AbbVie keeps on their website annually. Any big announcement by AbbVie may come in the leadup to their presentation at this event. I am not certain that they would have had enough time to analyze the Androgel 1.62% data. Therefore, an announcement related to a strategic partnership with ANIP and the readout of LibiGel trial results could become pubic at his time.
American College of Cardiology – Annual Scientific Session & Expo runs from March 4 to 6, 2023. This is more likely the event that AbbVie presents the Androgel 1.62% results. LibiGel results should also be presented at this event.
If I knew, with certainty, that AbbVie was partnering with ANIP, I would consider it a Strong Buy, however, Merck may be in play. Therefore, AbbVie is currently a Buy. Investors may want to consider acquiring options, with a strike date beyond March 6, 2023, on AbbVie and Merck, by then all the data and partnerships, if any, should be public knowledge.
Considering the strong possibility that an NDA has been, or will be, filed for female testosterone, ANIP is currently a Strong Buy. ANIP, with such small number of outstanding shares, does not take much trading activity to move the needle. Therefore, PPS would climb quickly on NDA and/or partnership announcements, which could come in the lead up to the JP Morgan Healthcare Conference.


Be the first to comment