FG Trade
A Quick Take On Structure Therapeutics Inc.
Structure Therapeutics Inc. (GPCR) has filed to raise $119 million in an IPO of its American Depositary Shares representing underlying ordinary shares, according to an S-1 registration statement.
The firm is a clinical-stage biopharma developing new therapeutics for the treatment of serious chronic diseases.
GPCR has significant operations in China and Australia.
When we learn more about the IPO from management, I’ll provide a final opinion.
Structure Therapeutics Overview
South San Francisco, California-based Structure Therapeutics Inc. was founded to develop G-protein-coupled receptors, called GPCR, as small molecule therapies that promise to overcome the limitations of biologics and peptide therapies that target the GPCR-family of receptors.
Management is headed by Chief Executive Officer Raymond Stevens, Ph.D., who has been with the firm since February 2019 and was previously the founder of the Bridge Institute at the University of Southern California and has been a long-time professor of Biology at The Scripps Research Institute.
The firm’s lead candidate, GSBR-1290 is being developed for the treatment of Type 2 diabetes and has initiated a Phase 1b proof-of-concept study for this form of diabetes and obesity.
Management expects topline data from its Phase 1b and Phase 2a study sometime in the second half of 2023.
Below is the current status of the company’s drug development pipeline:
Company Pipeline Status (SEC)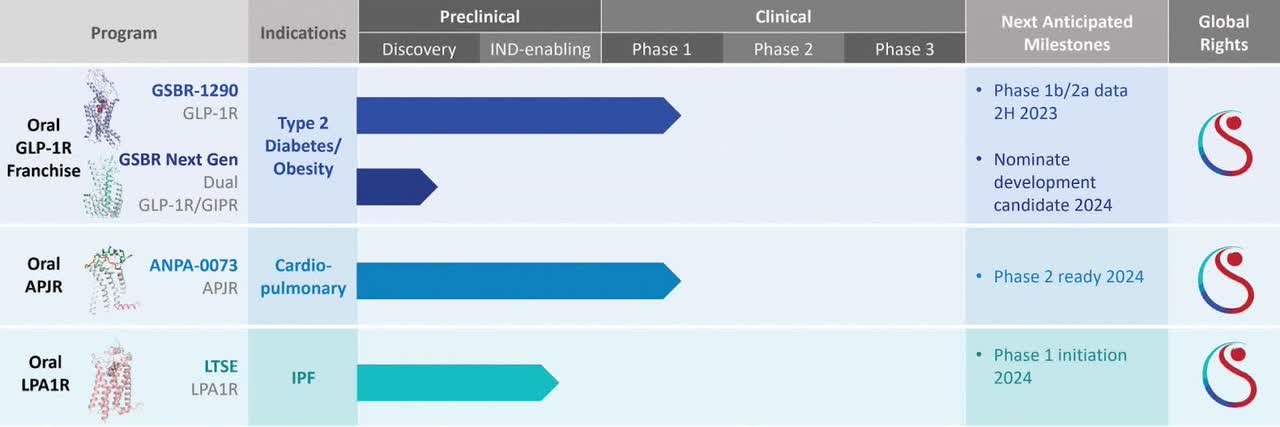
Structure has booked fair market value investment of $200 million as of September 30, 2022, from investors including ERVC Healthcare, F-Prime Capital Partners, Qiming, XX-I SHT Holdings, Biotechnology Value Fund, Sequoia Capital China and Deep Track Biotechnology.
Structure’s Market & Competition
According to a 2022 market research report by Allied Market Research, the global diabetes therapeutics market was an estimated $118 billion in 2020 and is forecast to reach $318 billion by 2030.
This represents a forecast CAGR (Compound Annual Growth Rate) of 10.4% from 2021 to 2030.
Key elements driving this expected growth are the growing incidence of diabetic conditions among an aging global population.
Also, the International Diabetes Federation reported that an estimated 537 million adults are currently living with diabetes.
That estimate is expected to reach 643 million by 2030 and 783 million by 2045.
Major competitive vendors that provide or are developing related treatments include:
-
Pfizer (PFE)
-
Eli Lilly & Company (LLY)
-
Qilu Regor Therapeutics
-
Jiangsu Hansoh Pharmaceutical Group
-
Boehringer Ingelheim
-
Altimmune (ALT)
-
Carmot Therapeutics
-
Sciwind Biosciences
-
CohBar
-
Apie Therapeutics
-
BioAge Labs
-
Others
Structure Therapeutics’ Financial Status
The firm’s recent financial results are typical of a clinical-stage biopharma in that they show no revenue and significant R&D and G&A costs associated with its drug development efforts.
Below are the company’s recent financial results:
Statement Of Operations (SEC)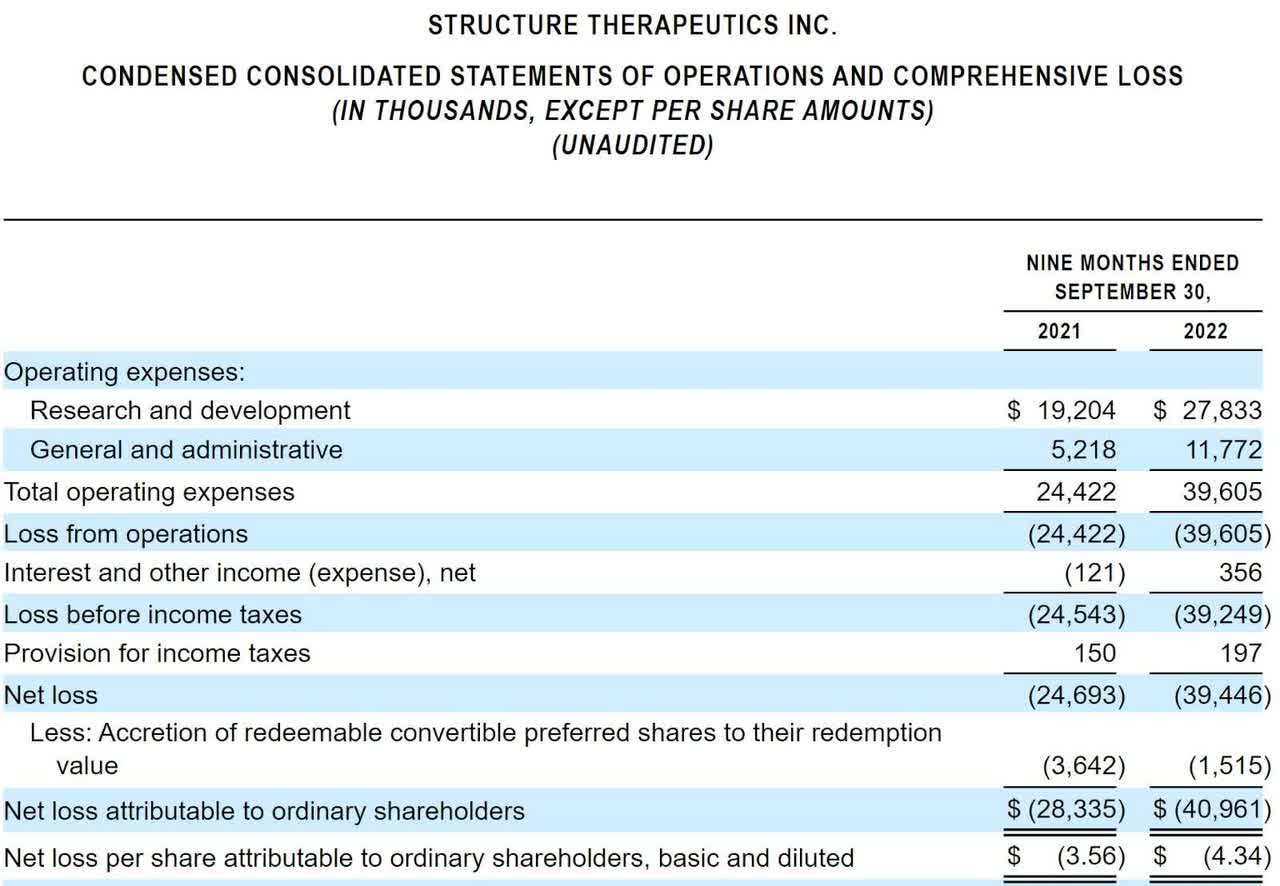
As of September 30, 2022, the company had $28.1 million in cash and $12.3 million in total liabilities.
Structure Therapeutics IPO Details
Structure intends to raise $119 million in gross proceeds from an IPO of its American Depositary Shares representing underlying ordinary shares, although the final figure may vary.
No existing shareholders have indicated an interest in purchasing shares at the IPO price, although this element may become a feature of the IPO if disclosed in a future filing.
Management says it will use the net proceeds from the IPO as follows:
to advance the development of our GLP-1R franchise, including the completion of a Phase 1b MAD study and Phase 2a proof-of-concept study, and next generation GLP-1R candidates including dual GLP-1R/GIPR agonists;
to advance the development of our [i] APJR agonist program, including through the initiation of a Phase 1 formulation bridging PK study as well as additional preclinical development studies in IPF and PAH, and [ii] LPA1R antagonist program, including preclinical development and initiation of our first-in-human study in IPF; and
the remaining proceeds to fund other research and development activities and general corporate purposes, which we expect will include the hiring of additional personnel, capital expenditures and the costs of operating as a public company.
(Source – SEC)
Management’s presentation of the company roadshow is not available.
Regarding outstanding legal proceedings, management says the firm is not subject to any material legal action or governmental investigation.
The firm has a number of wholly-owned subsidiaries in Australia ‘to conduct various preclinical and clinical activities for our product and development candidates in Australia.’
Structure also has material operations in China.
Listed bookrunners of the IPO are Jefferies, SVB Securities, Guggenheim Securities and BMO Capital Markets.
Commentary About Structure’s IPO
GPCR is seeking U.S. public capital market funding to advance its lead and other programs through and into clinical trials.
The firm’s lead candidate, GSBR-1290, is being developed for the treatment of Type 2 diabetes and has initiated a Phase 1b proof-of-concept study for this form of diabetes and obesity.
Management expects topline data from its Phase 1b and Phase 2a study to be available in 2H 2023.
The market opportunity for treating diabetes is large and expected to grow at a reasonably high rate of growth in the coming years.
However, the industry has strong competition among major pharmaceutical firms.
Management hasn’t disclosed any major pharma firm collaboration but does have a discovery collaboration with contract manufacturer Schrödinger.
The company’s investor syndicate includes a number of institutional venture capital firms, so the firm is well-capitalized.
Jefferies is the lead underwriter and its sole IPO led by the firm over the last 12-month period has generated a return of negative (0.6%) since its IPO. This is a mid-tier performance for all major underwriters during the period.
A potentially significant risk to the company’s outlook is the uncertain future status of Chinese company stocks in relation to the U.S. HFCA act, which requires delisting if the firm’s auditors do not make their working papers available for audit by the PCAOB.
While the firm’s current auditors have made their working papers available for audit by the PCAOB, as the company says, ‘there is no guarantee that future audit reports will be prepared by auditors subject to inspection by the PCAOB…’
Prospective investors would be well advised to consider the potential implications of specific laws regarding earnings repatriation and changing or unpredictable Chinese regulatory rulings that may affect such companies and U.S. stock listings.
When we learn more about the IPO from management, I’ll provide a final opinion.
Expected IPO Pricing Date: To be announced.


Be the first to comment