coldsnowstorm
Relay Therapeutics, Inc. (NASDAQ:RLAY) is a clinical stage developer of precision medicines, that is, they are discovering medicines using supercomputers, as Evaluate phrases it. They have a platform called Dynamo, which, they say in their 10-K, uses “computational and experimental approaches designed to drug protein targets that have previously been intractable or inadequately addressed.” The pipeline looks like this:
Relay pipeline (Relay website)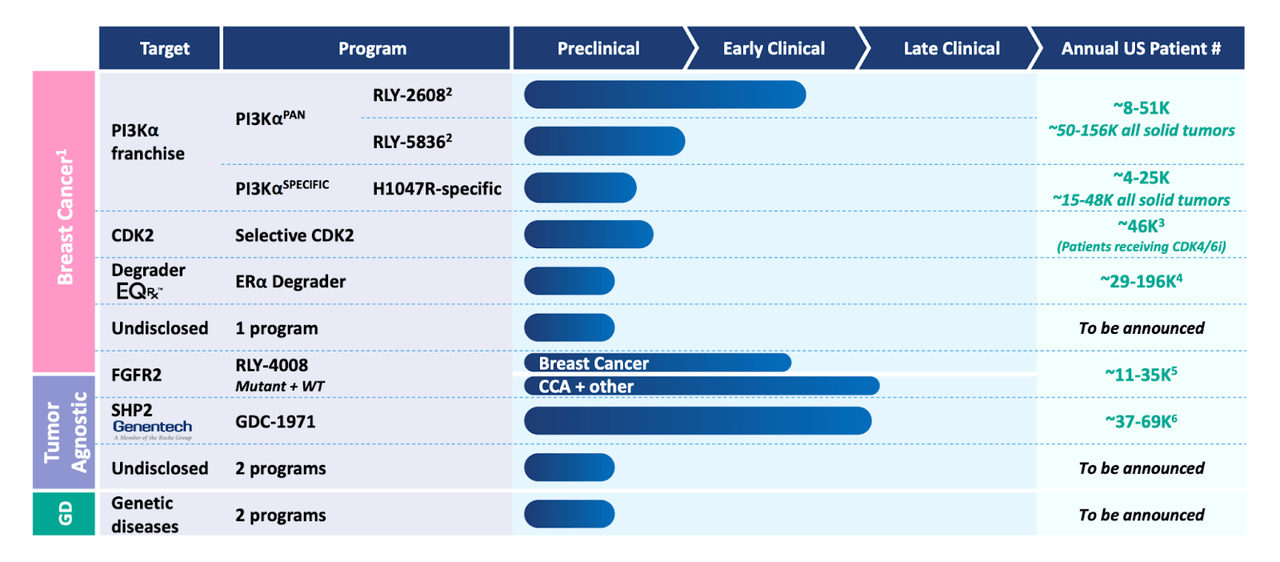
Among the company’s lead product candidates are RLY-4008, an oral small molecule inhibitor of fibroblast growth factor receptor 2 (FGFR2), which is in a first-in-human clinical trial for patients with advanced or metastatic FGFR2-altered solid tumors; RLY-2608, a lead mutant-PI3Ka inhibitor protein that targets phosphoinositide 3 kinase alpha; and RLY-1971, an oral small molecule inhibitor of protein tyrosine phosphatase Src homology region 2 domain-containing phosphatase-2 that is in Phase 1 trial in patients with advanced solid tumors. That last program has a tie-up with Genentech, a subsidiary of Roche (OTCQX:RHHBY).
RLY-4008 is the lead asset here, and it has data. 4008 is targeting FGFR2 in a highly selective manner. Other FGFR inhibitors are pan-FGFR, which creates certain limitations like low target inhibition, low tolerability and low efficacy, per the company:
FGFR limitations (Relay website)
RLY-4008 is a selective FGFR2 inhibitor. It was developed after the company observed that a certain segment of FGFR1 extends outward more frequently than the same segment in FGFR2. This enabled them to design a highly selective molecule. Interim data one cohort (FGFRi-Naïve Fusion+ CCA Patients at Pivotal Dose (70 mg QD)) from the ReFocus trial, which has a pivotal cohort among the four cohorts (FGFR2-fusion+ CCA without prior FGFRi), showed that 15 out of 17 patients had a treatment response, i.e., this was 88% ORR, where standard pan-FGFR inhibitors have shown less than half that number. The trial showed a 63% ORR across all dosage cohorts – 24 out of 38 patients.
The company noted at ESMO:
One pt treated at the RP2D had a near-complete response and subsequent tumor resection with curative intent. DOR is not yet mature, with majority of responses ongoing.
There were a few grade 3 AEs, and it appears that there was one discontinuation due to AE, but the molecule was generally well-tolerated. The company presents the main differences with a nice comparison graphic, see the image just above, and compare with the one below:
4008 Positives (Relay website)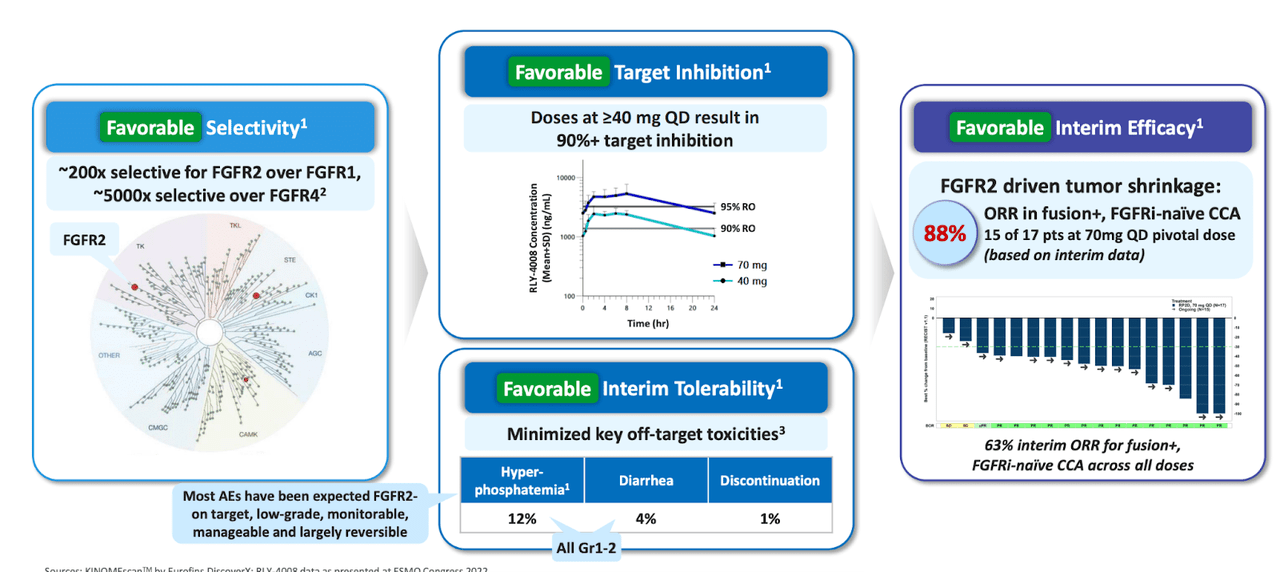
In 2023, they will have the following data milestones:
-
1H 2023: Full dose escalation data
-
2H 2023: Pivotal cohort full enrollment
-
2H 2023: Non-CCA expansion cohorts data.
The company noted in a press release in June that:
“End-of-phase 1 meeting with the U.S. Food and Drug Administration (FDA) resulted in alignment on the design of a single arm trial for pan-FGFR (FGFRi) treatment-naïve FGFR2-fusion cholangiocarcinoma (CCA) to potentially support accelerated approval.”
They have no other clinical data for any of the other self-owned programs. The Genentech partnered program has some data, but I will not discuss that data here. The Genentech partnership started in 2020. The key molecules were SHP2 and KRAS G12C inhibitors. Relay Therapeutics received $75 million upfront and an additional $25 million in near-term payments and $695 million in additional potential milestones, plus royalties on global net product sales. The company also has an R&D collaboration with D.E. Shaw Research, with whom they co-own a few patents and pending patent applications.
Financials
RLAY has a market cap of $2.47bn and a cash balance of $1.1bn, which includes the $300mn they raised through a secondary offering in September. Research and development expenses were $66.9 million for the third quarter of 2022, while general and administrative expenses were $16.1 million. At that rate, given the huge cash reserve, Relay Therapeutics, Inc. has a cash runway of more than 10 quarters.
Bottom Line
Although Relay Therapeutics, Inc. touted the theoretical low toxicity of its selective FGFR2 inhibitor, the trial data did not bear out that claim. There were annoying toxicities in the trial, including mouth and nail toxicities as well as hyperphosphatemia. A twice-daily dose had to be abandoned, and the high valuation vis-a-vis the so-so tox profile raises concerns. On the positive side, the drug surely works, and works much better than rival molecules; but this is only interim data. Safety data is also highly competitive.
Thus, I like Relay Therapeutics, Inc. for its novel science, but I will wait for a correction before taking a call.
Editor’s Note: This article discusses one or more securities that do not trade on a major U.S. exchange. Please be aware of the risks associated with these stocks.
About the TPT service
Thanks for reading. At the Total Pharma Tracker, we offer the following:-

Our Android app and website features a set of tools for DIY investors, including a work-in-progress software where you can enter any ticker and get extensive curated research material.
For investors requiring hands-on support, our in-house experts go through our tools and find the best investible stocks, complete with buy/sell strategies and alerts.
Sign up now for our free trial, request access to our tools, and find out, at no cost to you, what we can do for you.



Be the first to comment