BlackJack3D/iStock via Getty Images
Investment Summary
Equity markets continue to reprice med-tech stocks with heavy downside coming into the new year. This kind of pattern was well observed in Maravai LifeSciences Holdings, Inc. (NASDAQ:MRVI) following our last publication on the company back in August [we encourage investors to read it, click here]. Since then, we’ve seen investors re-rate the stock more than 44% to the downside to its current market cap. Last time, we said that valuations were a concern for the company, and that there could be more compelling opportunities elsewhere. Nevertheless, return on its capital investments was a standout. We noted this strength in return on invested capital continued into its latest set of numbers.
From that point, there’s been several developments concerning the transition of its CEO. Whilst we believe MRVI is a quality company in the long-term, right now, the investment metrics don’t add up to advocate for a strong buy. We note the stock’s attractive current discounted valuations, and this could be an upside risk looking ahead. But this doesn’t translate into EPS upside, and with potential overhang from the CEO transition, I’m back here today to reiterate our hold position on MRVI, price target $13.90.
Exhibit 1. Heavy selloff in MRVI equity across FY21′-22′. Question is, can the new management turn this around?
New CEO transition: Overhang, or turning point?
In early October 2022, William “Trey” Martin III was appointed as CEO of MRVI. However, by mid-October three subsidiaries of Danaher Corporation – two of which had previously employed Martin – filed a lawsuit against both Martin and MRVI in the Delaware Court of Chancery, alleging that Martin had breached a non-competition agreement. Carl Hull, MRVI’s co-founder was reinstated as interim CEO. As a result, a temporary restraining order was issued against Martin, preventing him from working for MRVI until the matter was sorted.
This was later resolved in December, with a confidential settlement reached between Martin, MRVI, and the Danaher entities. Under the settlement, Martin agreed to limit his responsibilities at MRVI, and refrain from involvement in the company’s Nucleic Acid Production Segment until July 2023. Consequently, he became President of MRVI’s biologics safety testing segment from December 5, FY22′, and is expected to assume the role of CEO on July 27 this year. Meanwhile, Christine Dolan, the current COO of the biologics safety testing Segment, will continue in her role and report to Martin. Hull will remain as the company’s executive chairman and interim CEO in the meantime.
We believe this to be a key point of consideration in the MRVI investment debate. It will either serve as a meaningful overhang to its share price, or a catalyst to re-rating once Martin steps into the role of CEO. From there, it will be up to Martin to earn his stripes, and we encourage investors to remain diligent on monitoring updates around his transition. As mentioned, should he convert on a new route of growth for the company, this could translate to share price appreciation.
Q3 earnings run-down: CleanCap headwinds clearly evident
We noted that MRIV reported Q3 total revenue of $191 million on adjusted EBITDA of $133 million [69% margin] and pulled this down to adjusted EPS of $0.37. Looking at this in further depth, the adjusted EBITDA compared to $155 million in Q3 FY21′, with the bolus of reconciliation from GAAP EBITDA to due to movements in stock-based compensation. Net-net, the total reconciliation amounted to ~6% off the GAAP EBITDA number for Q3.
It’s also worth noting that the nucleic acid production (“NAP”) business saw a 6% year-over-year decrease in revenue, due in part to a strong Q3 2021 comparison with a “large non-COVID order” from a single customer that was beginning a clinical trial. Moreover, we also observed a 400bps YoY decline in COVID-related CleanCap revenue in the NAP division to $127mm, compared to $131mm in Q3 2021 [more on this segment later]. It pulled in $134mm in adjusted EBITDA for the quarter.
Turning to the biologics safety testing business, it experienced intermittent headwinds in China, resulting in a 100bps drop in revenue compared to the same period in the previous year. This resulted in adjusted EBITDA of $13mm for the period.
Looking down the P&L, R&D expenses in Q3 were over $5 million, up from ~$2 million spent in same time last year, as the company continues its expansion focus on the CleanCap franchise. As such, operating income pulled in to $117 million, resulting in an operating margin of 61%. MRVI realized a free cash inflow of $67.9 million in Q3 2022 [$119 in adjusted terms], pushing its on-balance sheet cash balance to $617 million as of the end of the quarter, a sequential increase of $67 million from Q2.
Despite this, ended Q3 with long-term debt of $540 million, with net CapEx of just $14mm. Management expects net capital expenditures for 2022 to drift between $50-$55 million. The breakdown of deployment works out to be ~66% long-term assets and the remainder to its traditional fixed assets. The company has a cap on its cash-based interest rate at 6.5% through an interest rate cap contract.
These are important takeouts in our opinion. Especially when taking a deeper look at MRVI’s profitability measures. As you’ll see below, the company’s free cash flows and return on invested capital (“ROIC”) has followed a similar trajectory since Q1 FY222. In fact, the company’s return on its investments has dwindled to ~10% from highs of nearly 20% a year ago. Still, these are fairly solid numbers in terms of quarterly ROIC, still above the quarterly WACC figure of 8.82%. We’d be interested to see how these numbers progress in the company’s full-year earnings. This gives us more confidence in MRVI’s ability to fund its own growth initiatives looking ahead.
Exhibit 2. Despite substantial pullback in profitability and ROIC, this is well explained from lower Covid-19 revenues and recognized in its financial statements. Moreover, ROIC still outpaces the WACC hurdle for now.
Data: Author, MRVI SEC Filings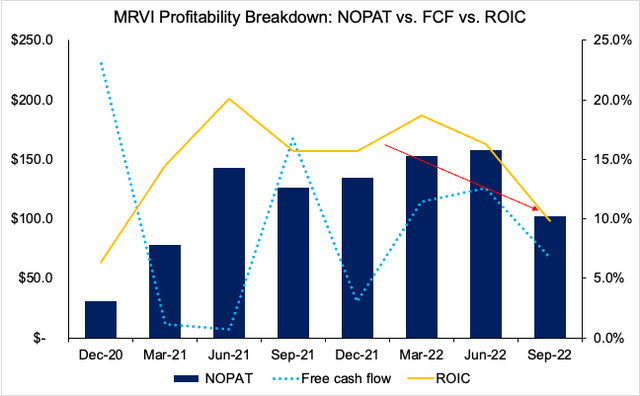
A further look at the CleanCap headwinds and guidance
Recent investor concerns have centred around the longevity of its COVID-related CleanCap revenues into 2023 and beyond, given that approximately two-thirds of the company’s revenue for 2022 is expected to be derived from the utilization of CleanCap in COVID-19 vaccines. However, the vaccine industry is currently experiencing a significant degree of uncertainty, and the uptake of the new bi-valent booster vaccines has been relatively low, with only 19 million individuals in the U.S. having received the booster dose as of October 19, FY22′, according to management’s language on the earnings call. As a result, MRVI anticipates that COVID-related vaccine production will likely decrease by 50-66% from 2022 levels, leading the company to initially estimate that its COVID-related CleanCap revenues for 2023 will fall within a range of $200-300 million.
However, MRVI has recently revised this estimate to the downside due to a lack of binding commitments and long-range forecasts from major customers, along with the assumption that customers currently have excess inventories on hand. Each of these factors is expected to negatively impact revenue in FY23 – particularly in the early part of the year as these inventories are depleted. As a result, MRVI now estimates that its CleanCap COVID revenue for FY23′ will be ~$100 million, with limited shipments expected in H1. Internally, the company is also planning for a c.$100 million annualized run rate for COVID-related CleanCap revenue in FY24 and beyond.
And with that, it’s worthwhile discussing MRVI’s FY22 guidance numbers. It has rebased its revenue projections for the full-year within a range of $880 million-$890 million. Curiously, this including an estimate of $600 million-$605 million of revenues related to CleanCap demand for COVID-19 vaccines. Overall, these numbers represent a 100bps alteration at the midpoint of range. Moreover, it implies a midpoint of approximately $282 million for MRVI’s non-COVID CleanCap business, calling for ~17% YoY.
Management also forecast 3-5% YoY growth for the biologic safety testing segment and 35-40% growth in the NAP business. It also maintained FY22′ EBITDA guidance, to a range of $650 million-$660 million. It expects to pull in adjusted EPS in a range of $1.76 to $1.80 per share.
Valuation and conclusion
It’s first worth noting that MRVI is attractively rated using Seeking Alpha’s quantitative factor gradings [Exhibit 3]. It is rated highly in terms of valuation, giving more confidence in advocating against a sell.
As such, the stock is trading at 10.3x forward earnings [GAAP estimates], and even cheaper at 7.8x non-GAAP estimates. These are discounted multiples that could offer potential valuation upside, however, could also compress forward looking price objectives.
Consensus also has the company to deliver $1.35 in forward GAAP EPS, and assigning this to the 10.3x multiple to this derives a price target pf $13.90, offering negligible upside.
If looking at FY23′ estimates however, GAAP EPS estimates are 0.35, and assigning the 10.3x multiple registers an objective of just $3.60. This is a downside risk that must be factored into the investment debate, and further confirms a neutral view.
Exhibit 3. MRVI Seeking Alpha Factor Grading showing that MRVI’s starting valuation could potentially be attractive.
Data: Seeking Alpha, MRVI Quote page, see “Factor Grades”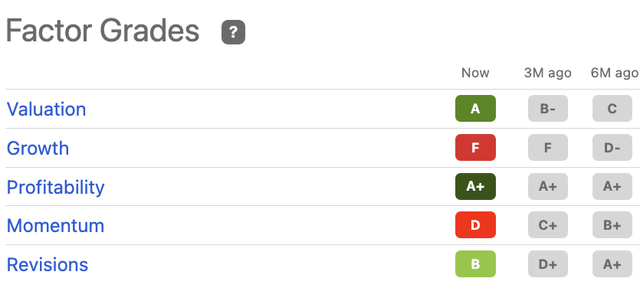
Net-net, there isn’t substantial evidence to advocate MRVI as a buy right now. There are many unanswered questions. However, there’s also scope for investors to remain constructive on this stock. It is trading at a heavy discount to peers, and it has the ROIC to fund its future growth initiatives. These are two points that could stand out into the future. We’re looking to the company’s Q4 and FY22 numbers to make a more informed decision here, and this could extend until the insertion of the new CEO in ~July FY23′. Rate hold.


Be the first to comment