
metamorworks
May those who follow their fate be granted happiness; may those who defy it be granted glory”― Mizuo Shinonome
Today, we take a look at a small gene therapy concern for the first time. The shares have seen some recent purchases by a beneficial owner. An analysis follows below.
Seeking Alpha
Company Overview:
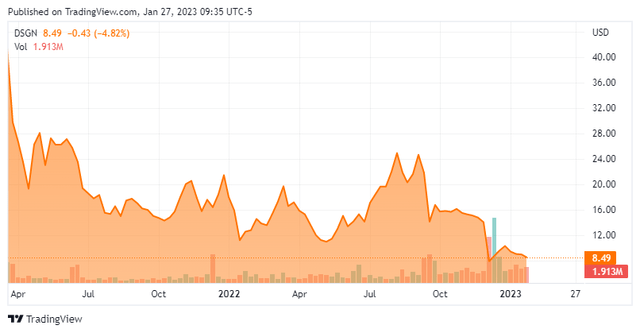
Design Therapeutics, Inc. (NASDAQ:DSGN) is a Carlsbad, California early clinical-stage biopharmaceutical concern focused on the development of gene targeted chimera therapies for diseases caused by inherited nucleotide repeat expansion [NRE] mutations. The company has one asset in the clinic for the treatment of Friedreich’s ataxia [FA] and two in the preclinic for the treatment of Fuchs endothelial corneal dystrophy (FECD) and myotonic dystrophy type-1 (MD1). Design was founded in 2017 and went public in March 2021, raising net proceeds of $254.3 million at $20 per share. The stock is around $8.50 per share, translating to an approximate market cap of $475 million.
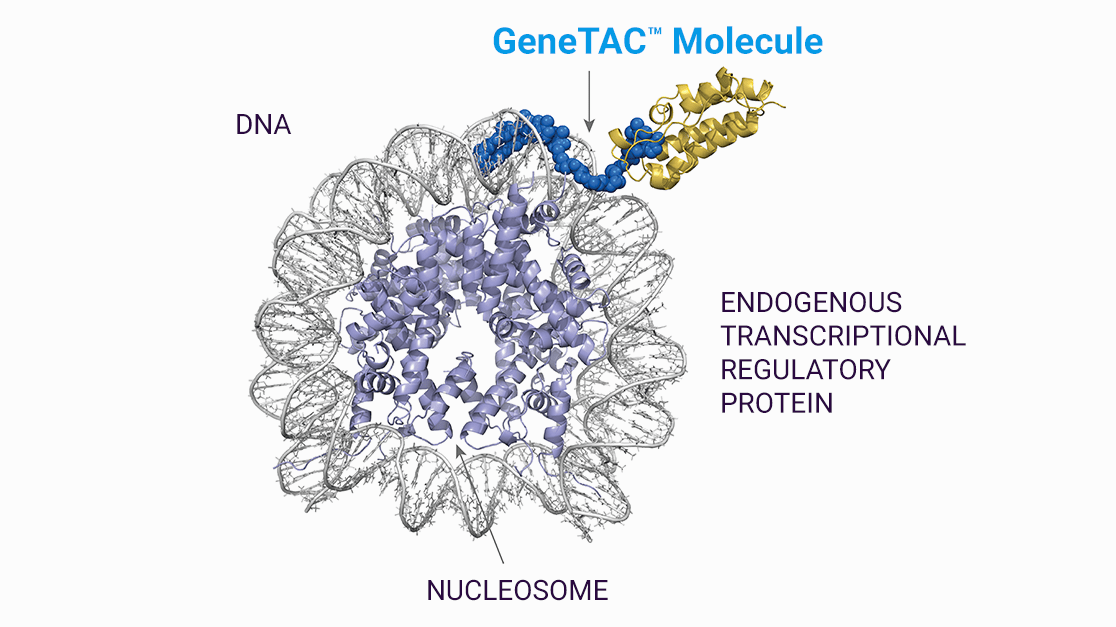
GeneTAC Platform
Individuals with NRE mutations are born with hundreds to thousands of excess nucleotide repeats in their distorted gene. NRE has been identified as the underlying cause of over 40 degenerative diseases with no currently approved remedies targeting them.
Company Website
Specific DNA sequences of a gene can generate RNA through a process called transcription. In turn, the RNA is employed as a template to produce proteins that are responsible for cellular function in a process called translation. Combined, these two operations are responsible for gene expression. Individuals with NRE in the DNA are subject to different alterations of transcription, resulting in either insufficient production of a crucial protein or a cascade of downstream toxicity and cellular dysfunction arising from a misprocessed RNA transcript.
To address these NRE diseases, Design has created a development platform dubbed GeneTAC. It designs heterobifunctional compounds, meaning they are comprised of two moieties, each with a unique function, connected via linker. On one end is the DNA-targeting moiety that is designed to recognize and pursue a specific nucleotide repeat sequence. On the other end is a ligand moiety that is designed to interact with the endogenous transcription-regulating proteins and modulate the transcription machinery in the cell by either dialing up or dialing down gene expression. This ligand moiety can be tailored to either restore transcription or reduce toxic gene product levels.
Pipeline
To date, GeneTAC has produced one clinical program, with two others in the preclinic.
DT-216. The company’s lead asset is DT-216, which is designed to modify FA, a dreadful monogenic, autosomal recessive progressive disorder where 95% of cases are the result of homozygous guanine-adenine-adenine [GAA] triplet repeat expansions in the first intron of the FXN gene on chromosome nine, which encode the mitochondrial protein FXN. Symptoms typically present around puberty. They include difficulty walking, sensation loss in the arms and legs, scoliosis, hearing and vision loss, and diabetes mellitus. By early adulthood, most patients are wheelchair bound, with loss of life usually occurring from hypertrophic cardiomyopathy (60%) in their late 30s. It afflicts more than 5,000 living individuals in the U.S. and more than 20,000 in Europe with an incidence of 1 in 40,000 to 50,000 births.
The number of repeats range from up to 30 GAA triplets in healthy individuals to ~300-1,000+ in FA patients, which retard their ability to produce the FXN protein, resulting in levels approximately one-quarter of normal. Interestingly, heterozygote carriers typically possess half the normal FXN levels and function asymptomatically, meaning a doubling of FXN protein is potentially all that is necessary to provide therapeutic benefit.
DT-216 consists of a DNA-binding moiety designed to connect to the expanded GAA sequence in the first intron of the FXN gene and a ligand moiety designed to recruit an endogenous transcriptional elongation complex to unclog the transcriptional machinery; thus restoring FXN production to normal (or at least therapeutic) levels. In a 39-adult-patient Phase 1 placebo-controlled, six-cohort, single ascending dose (SAD) study that initiated in March 2022 and readout on December 7, 2022, DT-216 (in all cohorts 100mg and above (n=15)) demonstrated a statistically significant increase in FXN mRNA at 24 hours ranging from 1.24 to 2.62-fold. The therapy was well-tolerated with no severe adverse events reported.
However, there was no change in FXN protein expression at 24 hours, a seemingly unrealistic expectation considering the SAD nature of the study and the truncated window. Nonetheless, investors were expecting more and sold in the subsequent trading session, removing one-third of its market cap for a close at $8.46 a share. That said, Design isolated pre-treatment peripheral blood mononuclear cells from trial patients and treated them with 100 nanometers of DT-216 for 60 hours, which resulted in a doubling of FXN protein.
DT-216 is also undergoing evaluation in an ongoing multiple ascending [MAD] dose Phase 1 trial designed to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of three weekly doses of DT-216 in adult patients with FA. The main objective is to determine a dose required to achieve sufficient DT-216 biodistribution in muscle for a potentially registrational Phase 2 trial expected to initiate in 2H23. The first MAD cohort of 100mg was dosed as of December 2022. Design anticipates dosing at least three cohorts before reporting data in mid-2023. DT-216 has received Fast Track designation from the FDA.
Designation notwithstanding, the company’s lead candidate may have some catching up to do as Reata Pharmaceuticals (RETA) has a PDUFA date of February 28, 2023 for its once-daily, oral compound omaveloxolone, which activates transcription factor Nrf2 to restore mitochondrial function in FA patients. Also, data are expected in 2Q23 from a potentially registrational FA trial for PTC Therapeutics’ (PTCT) inhibitor of energetic and oxidative stress pathway regulator 15 lipoxygenase (vatiquinone). Privately held Retrotope’s synthetic linoleic acid RT001 is undergoing evaluation in a pivotal Phase 2/3 trial, while Larimar Therapeutics (LRMR) and privately held Minoryx Therapeutics have earlier-stage FA programs in the clinic.
DT-168. Design’s leading preclinical asset is DT-168, which is an eyedrop designed to treat FECD, a bilateral, progressive, genetic corneal disease characterized by deterioration of endothelial cells, development of cornea-irritating fluid (guttata), and subsequent decreased visual acuity. FECD afflicts more than one million in the U.S. with ~4% of Americans over 40 years of age (~six million) exhibiting symptoms of the disease. Nearly three-quarters of all cases are caused by a CTG trinucleotide repeat expansion within the transcription factor 4 gene, leading to the formation of toxic RNA foci, global splicing dysregulation, and the aforementioned endothelial cell deterioration and loss. Corneal transplantation is the only approved procedure to restore vision, which occurs in 10%-20% of FECD patients. After demonstrating promise in the preclinic, Design anticipates filing an IND in 2H23.
The company also has a program targeting DM1, a monogenic, autosomal dominant disease characterized through a myriad of manifestations – including myotonia, muscle weakness, cardiac arrhythmias, fatigue, GI issues, cataracts, and cognitive impairment – resulting from damage to skeletal, cardiac, and smooth muscles. It is caused by an increase in the number of CTG triplet repeats found in the 3’ non-coding region of the DMPK gene. However, an IND is not anticipated to be filed until 2024.
Balance Sheet & Analyst Commentary:
To advance DT-216 through the clinic, as well as DT-168 and its DM1 asset into the clinic, Design held cash and investments of $344.2 million, providing it a cash runway through YE24, even though the pace of burn should increase meaningfully from its current ~$12 million per quarter. The company has no debt.
With one glaring exception, the Street is upbeat on Design’s prospects, albeit a little less so after the December 7th trial data. Analysts have four outperform ratings against one sell with their median price target falling from $29 to $22 post-readout. Goldman Sachs analyst Madhu Kumar’s bearish take is instructive. He suggests that natural history studies provide little support that FXN levels will alter FA disease course, pointing to the success of high-dose nicotinamide in raising FXN levels but with no therapeutic benefit to FA patients.
A good portion of the boardroom disagrees with Mr. Kumar’s concerns. CEO & President Joao Siffert (7,450 shares), Director Rodney Lappe (15,000), and Director Simeon George, representing the interests of beneficial owner SR One Capital (900,000), have all added to their positions post-data selloff.
Verdict:
The first readout for DT-216 was likely misinterpreted by the market as expectations for the development of FXN protein in 24 hours appear unrealistic. However, between the competition and the concerns articulated by Goldman Sachs analyst Kumar, DT-216 has a pretty narrow path to success. Even if the MAD trial yields an increase in FXN, DT-216 still needs to demonstrate therapeutic benefit – and if it does, it will not be on the market until FY25 at the earliest, providing Reata a two-year head start if its omaveloxolone is approved.
Design’s science appears sound, and the market is only valuing its platform and programs at ~$130 million when the balance sheet cash is backed out from its market capitalization. However, its first indication may be a tough needle to thread. As such, I will take a pass on any investment recommendation on this name.
Those who cannot own their choices will always be mastered by fate.”― Katy Rose Pool


Be the first to comment