Solskin
A Quick Take On Apogee Therapeutics, Inc.
Apogee Therapeutics, Inc. (APGE) has filed to raise $100 million in an IPO of its common stock, according to an S-1 registration statement.
The firm is a preclinical stage biopharma developing drug candidates for the treatment of inflammatory diseases.
APGE is highly capitalized but is still at a very early and high-risk stage of development, with a long development road still ahead of it.
When we learn more IPO details from management, I’ll provide a final opinion.
Apogee Overview
Waltham, Massachusetts-based Apogee Therapeutics, Inc. was founded to develop monoclonal antibody-based treatments for atopic dermatitis and chronic obstructive pulmonary disease.
Management is headed by Chief Executive Officer Michael Henderson, M.D., who has been with the firm since September 2022 and was previously Chief Business Officer of BridgeBio Pharma after holding several other senior positions there.
The firm’s lead candidate, APG777, is a monoclonal antibody targeting IL-13 for the treatment of atopic dermatitis, a skin condition affecting millions of people worldwide. The drug is also being investigated for the treatment of asthma.
APG777 is still in preclinical stage of development, and the company has filed for approval to begin Phase 1 safety trials in Australia.
Below is the current status of the company’s drug development pipeline:
Company Pipeline (SEC)
Apogee has booked fair market value investment of $179.7 million as of March 31, 2023, from investors including Fairmount Funds, Venrock, Deep Track Capital, FMR (Fidelity), RTW Investments and Paragee Holding.
Apogee’s Market & Competition
According to a 2022 market research report by Grand View Research, the global market for atopic dermatitis drugs was an estimated $12.7 billion in 2021 and is forecasted to reach $27.6 billion by 2030.
This represents a forecast CAGR (Compound Annual Growth Rate) of 9.0% from 2022 to 2030.
Key elements driving this expected growth are a strong pipeline of treatment options and a growing prevalence of diagnosed conditions due in part to increased handwashing resulting in ‘cracked and dry skin prone to infections among atopic dermatitis patients.’
Also, the chart below shows the historical and projected future growth trajectory of the atopic dermatitis treatment market in the U.S. from 2020 to 2030:
U.S. Atopic Dermatitis Market (Grand View Research)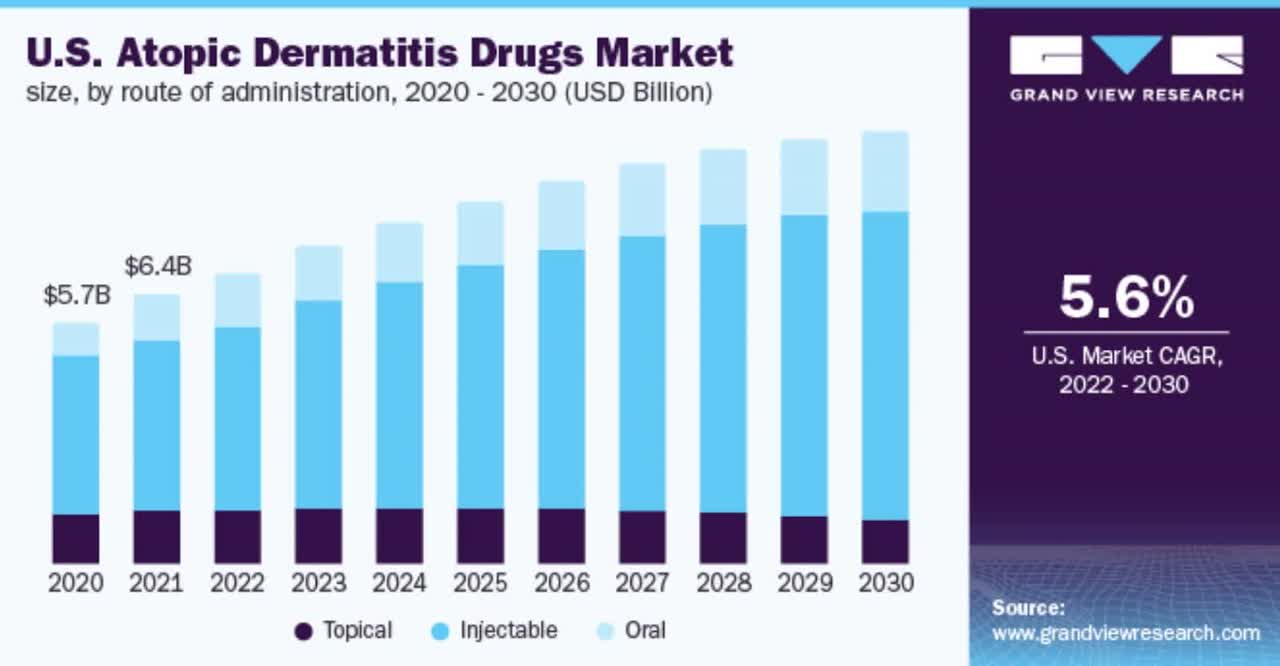
Major competitive vendors that provide or are developing related treatments include the following companies:
-
AbbVie (ABBV)
-
Johnson & Johnson (JNJ)
-
Novartis (NVS)
-
Pfizer (PFE)
-
Sanofi (SNY)
-
Galderma Laboratories
-
Eli Lilly and Company (LLY)
-
Viatris (VTRS)
-
Regeneron Pharmaceuticals (REGN)
-
LEO Pharma
-
Otsuka Pharmaceutical
-
Incyte (INCY)
-
Others
The company is also developing drug candidates for COPD, asthma and other serious medical conditions with large market demand.
Apogee Therapeutics’ Financial Status
The firm’s recent financial results are typical of a development stage biopharma in that they feature no revenue and material R&D and G&A expenses associated with its development efforts.
Below are the company’s financial results for the relevant periods:
Statement Of Operations (SEC)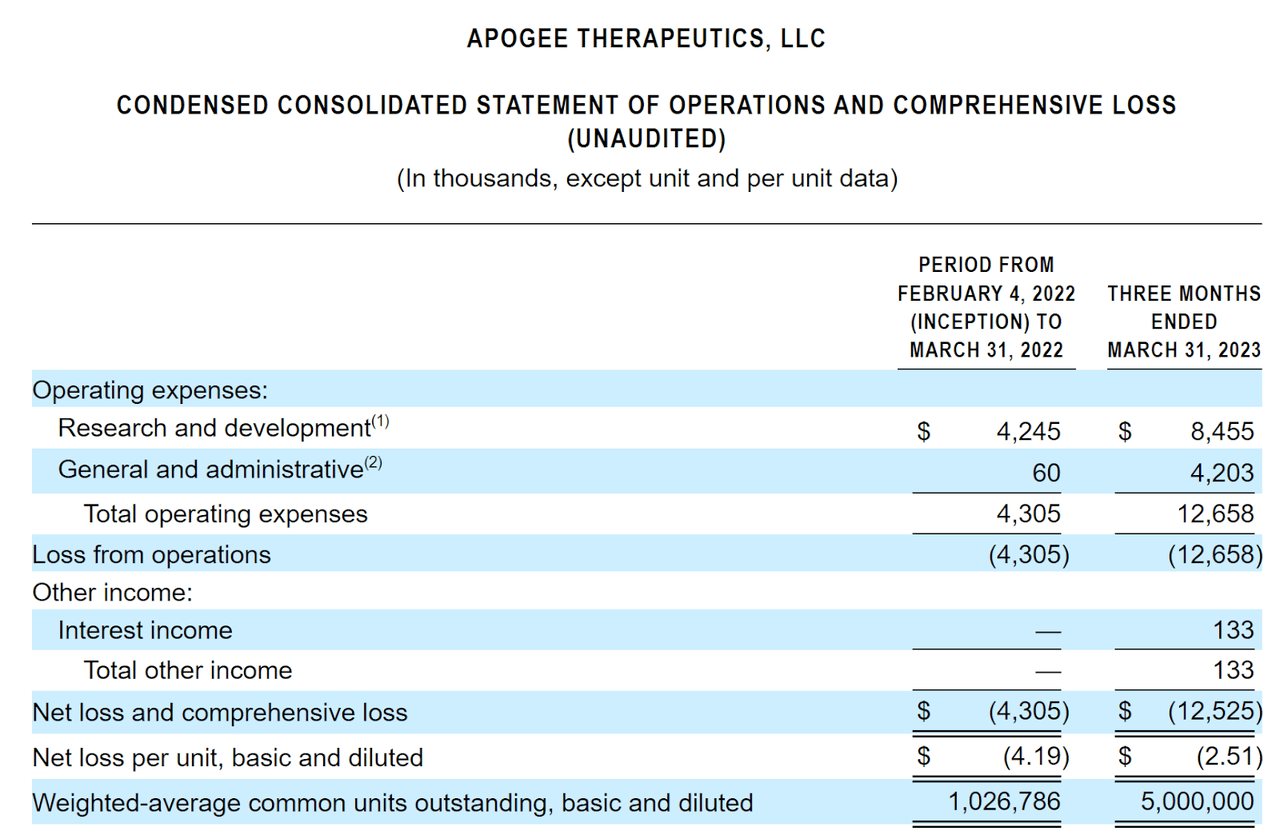
As of March 31, 2023, the company had $141.3 million in cash and $11.2 million in total liabilities.
Apogee Therapeutics, Inc. IPO Details
Apogee intends to raise $100 million in gross proceeds from an IPO of its common stock, although the final figure may differ.
The firm is spinning out from Paragon Therapeutics, a discovery company with whom Apogee considers itself a ‘related party.’
Apogee has two classes of common stock, voting and non-voting. The voting stock is being offered in this IPO.
The S&P 500 no longer admits firms with multiple classes of stock into its index.
No existing shareholders have indicated an interest in purchasing shares at the IPO price, although this element may become a feature of the IPO if disclosed in a future filing.
The firm is an ‘emerging growth company’ as defined by the 2012 JOBS Act and has elected to take advantage of reduced public company reporting requirements; prospective shareholders will receive less information for the IPO and in the future as a publicly-held company within the requirements of the Act.
Management says it will use the net proceeds from the IPO as follows:
Proposed Use Of Proceeds (SEC)
Management’s presentation of the company roadshow is not available.
Regarding outstanding legal proceedings, management said the firm is not currently a party to any legal proceedings that would have a material adverse effect on its business.
Listed bookrunners of the IPO are Jefferies, TD Cowen, Stifel, Guggenheim Securities and Wedbush PacGrow.
Commentary About Apogee’s IPO
APGE is seeking U.S. public capital market investment to fund its various drug candidate development and trial efforts.
The firm’s lead candidate, APG777, is a monoclonal antibody targeting IL-13 for the treatment of atopic dermatitis, a skin condition affecting millions of people worldwide. The drug is also being investigated for the treatment of asthma.
APG777 is still in preclinical stage of development, and the company has filed for approval to begin Phase 1 safety trials in Australia.
The market opportunities for the firm’s drug candidates are large and expected to grow as the global population ages and experiences greater incidences of health conditions.
The company’s IPO is a result of its collaboration partner Paragon Therapeutics from whom the firm is spinning out. Apogee has in-licensed certain IP for its IL-13, IL-14Ra and OX40L development programs.
The company’s investor syndicate includes well-known life science venture capital firms.
Jefferies is the lead underwriter, and the two IPOs led by the firm over the last 12-month period have generated an average return of 52.3% since their IPO. This is a top-tier performance for all major underwriters during the period.
Apogee is extremely well capitalized but is still at a very early and high-risk stage of development, with a long development road still ahead of it.
When we learn more IPO details from management, I’ll provide a final opinion.
Expected IPO Pricing Date: To be announced.


Be the first to comment