Bet_Noire
Tact is the knack of making a point without making an enemy.”― Isaac Newton.
Today, we put Seres Therapeutics, Inc. (NASDAQ:MCRB) in the spotlight. If all goes well, the company will garner its first FDA approval in the second quarter of this year. Will that bolster MCRB stock or is this news already priced into its shares? An analysis follows below.
Company Overview:
Seres Therapeutics, Inc. is a developmental concern focused on developing microbiome therapeutics which is based in Boston, MA. Currently, MCRB stock sells at around $5.50 a share and has an approximate market capitalization of $685 million.
December Company Presentation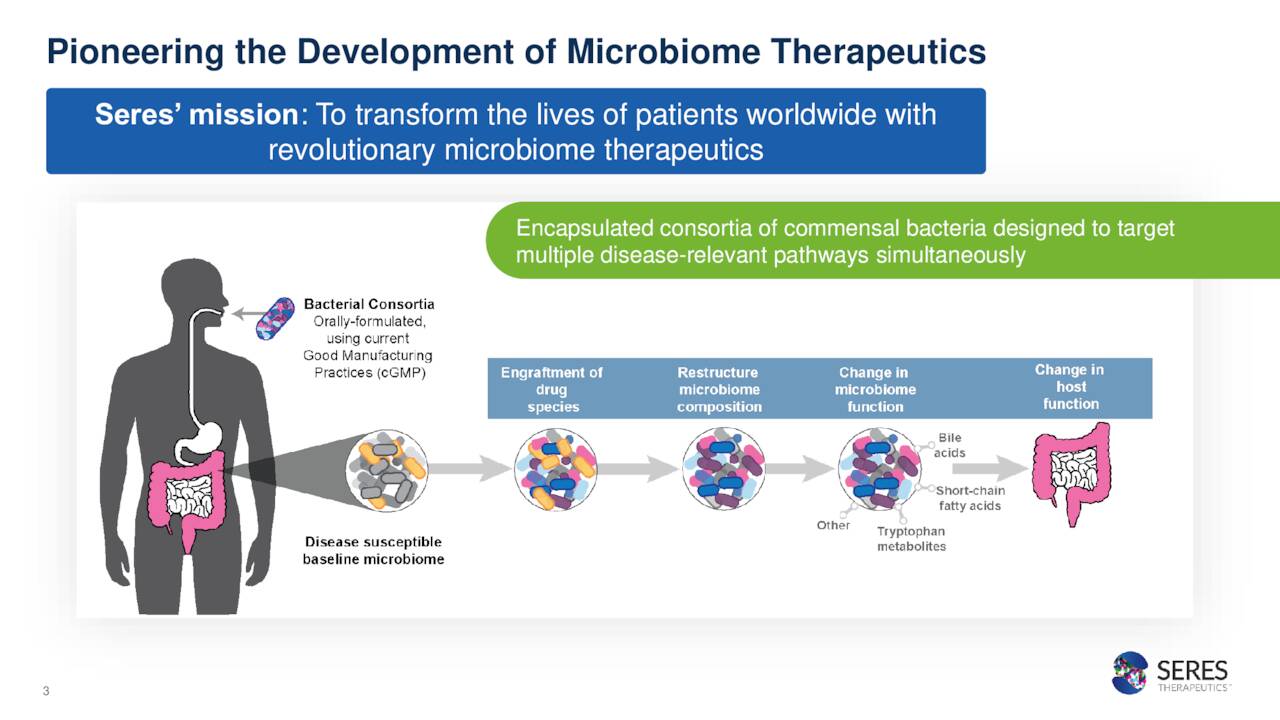
The company’s most advance compound in development is named SER-109. This an oral microbiome therapeutic candidate. The company has submitted a Biologic License Application or BLA around it as a treatment of clostridium difficile infection or CDI. CDI is short for the name of the germ that causes the infection: Clostridioides difficile.
December Company Presentation
This infection can affect anyone but it primarily affects those individuals that have recently spent some time in a hospital or in a long-term care facility, are 65 or older, have weakened immune systems and/or are taking, or have recently taken, antibiotics. The most common symptoms of CDI are watery diarrhea, nausea, stomach pain or cramps. These symptoms can last for days if not longer. If not treated right away, this infection can lead to serious medical problems. This is a large area of opportunity.
December Company Presentation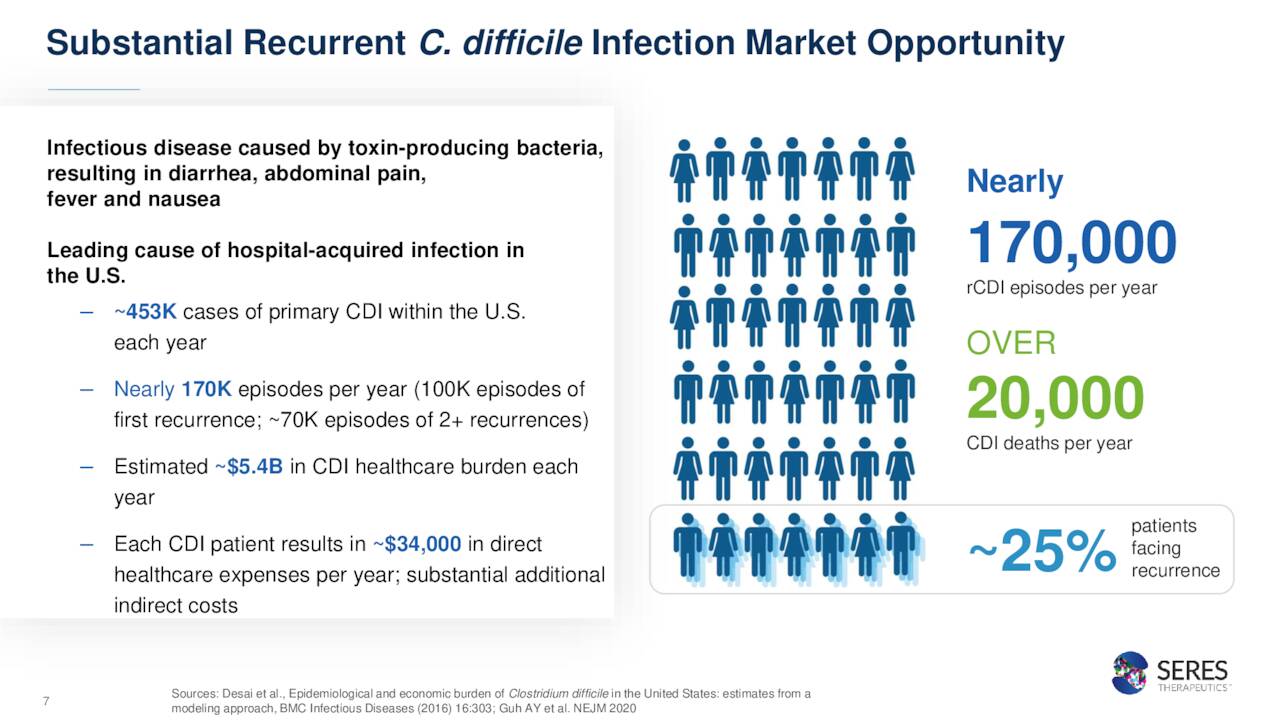
SER-109 is unique as it is a collection of live organisms. SER-109 comes in a pill form which can be digested at home. The PDUFA date for the application is April 26th, and it is partnered with Nestlé S.A. (OTCPK:NSRGF) in this effort. The FDA has not requested an Ad Comm Panel around this application and Phase 3 data was solid, so approval seems likely. The application also is getting a priority review thanks to its Breakthrough Therapy designation.
December Company Presentation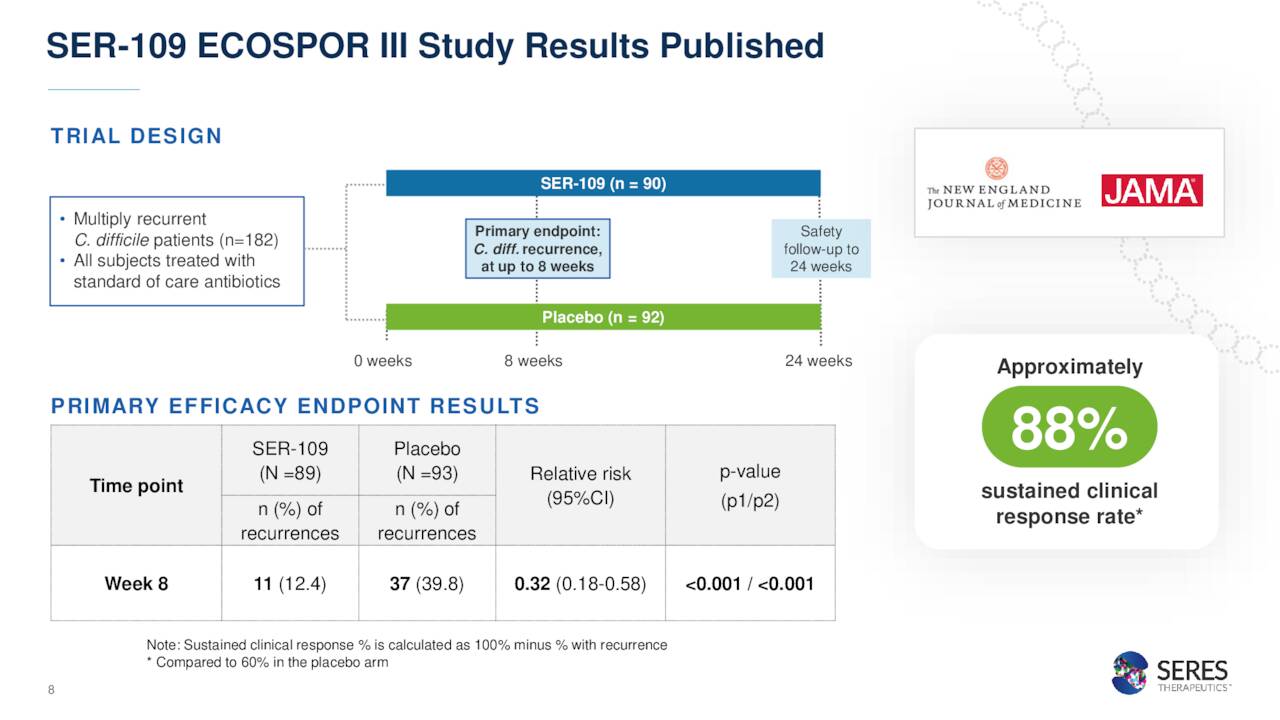
Seres is also developing a candidate called SER-155. This is a cultivated bacteria microbiome drug.
December Company Presentation
Currently this is in a Phase Ib clinical trial to reduce incidences of gastrointestinal infections, bloodstream infections, and graft versus host diseases in immunocompromised patients receiving allogeneic hematopoietic stem cell transplantation and solid organ transplants.
December Company Presentation
Seres Therapeutics, Inc. also has a couple of early stage candidates in development within this partnership focused around immune modulation. They will not be germane to this analysis, as the company is primarily focused on the approval and launch of SER-109 for the time being.
December Company Presentation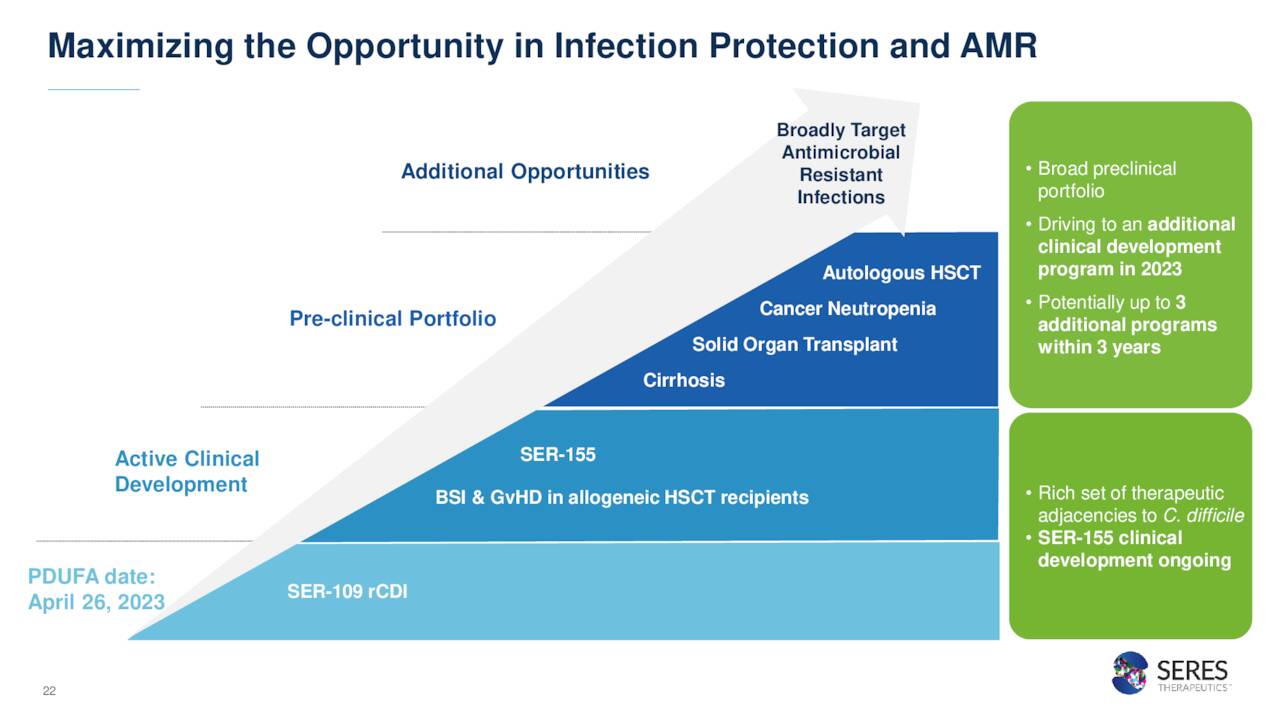
Analyst Commentary & Balance Sheet:
Over the past three months, as FDA approval looms large, Chardan Capital ($12 price target), Piper Sandler ($13 price target) and H.C. Wainwright ($25 price target) have all reiterated Buy/Outperform ratings on the stock.
Approximately seven percent of the outstanding float in the shares is currently held short. A beneficial owner bought nearly $30 million of a $100 million secondary offering the company did early last summer. An insider sold just less than $40,000 worth of shares last October. That is the only insider activity in the stock since late in 2020.
The company ended the third quarter with just over $230 million worth of cash and marketable securities on the balance sheet after posting an approximate net loss of $60 million in the quarter. It should be noted that the FDA approval of SER-109 will trigger a $125 million payment from Nestle to Seres and $225 million in milestone payments waits in wings. The company also has a 50/50 profit share with Nestle on commercialized sales.
Verdict:
Approval of SER-109 will be a big step forward using the microbiome to develop effective cures to a variety of afflictions. The microbiome is the collection of all microbes, such as bacteria, fungi, viruses, and their genes, which naturally live on our bodies and inside us. These entities protect us against pathogens, help our immune system develop, and enable us to digest food to produce energy. They are becoming a bigger focus for medicine as well as human wellness.
December Company Presentation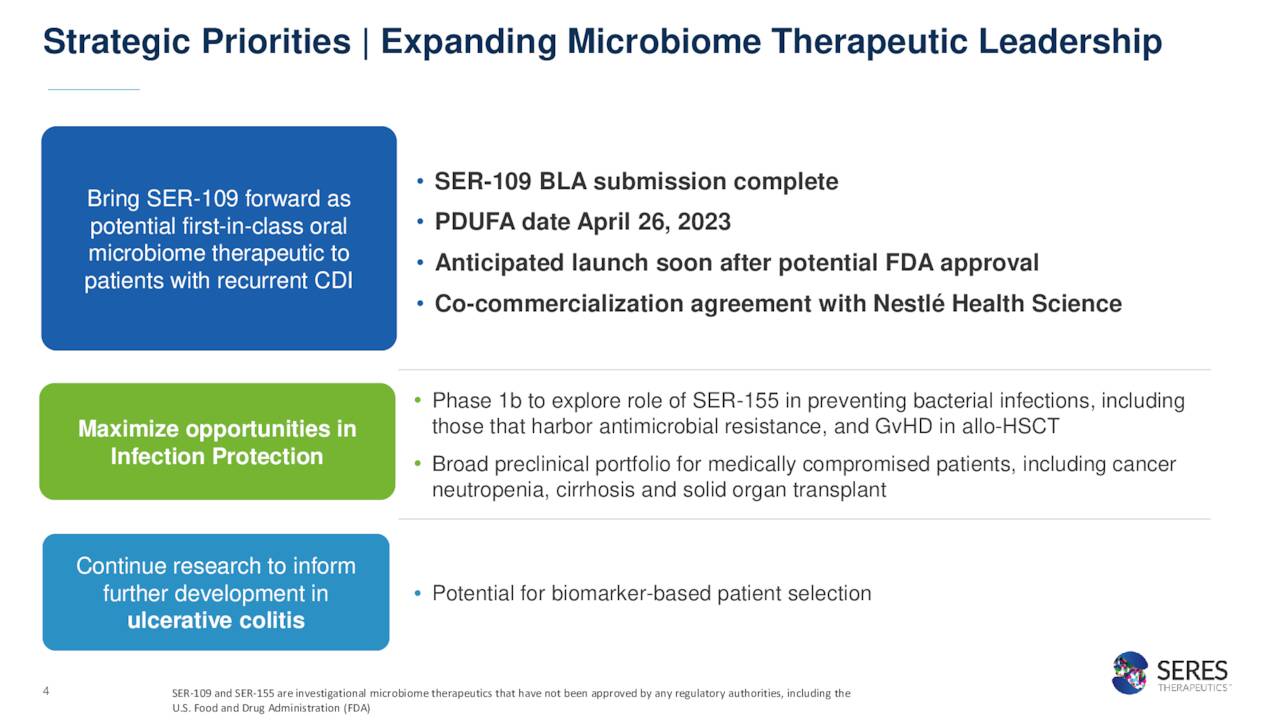
Nestle is a good partner to Seres Therapeutics, Inc. for this endeavor, as it is not a big pharma player but is instead focused on nutrition, health, and wellness. This seems a good fit for the areas Seres Therapeutics, Inc. is focused on developing products for. Cash burn will remain a concern, although the likely upcoming milestone payments do alleviate that worry for the near term. The pace of an initial rollout is also almost always tough to project (especially when pricing isn’t known yet) in this space. That said, there seems to be enough to the Seres Therapeutics, Inc. story to initiate a small “watch item” holding in MCRB, which I have recently done via covered call orders.
December Company Presentation
Force is all conquering, but it’s victories are short lived.”― Abraham Lincoln.
Editor’s Note: This article discusses one or more securities that do not trade on a major U.S. exchange. Please be aware of the risks associated with these stocks.


Be the first to comment