ipopba
People never lie so much as after a hunt, during a war, or before an election.”― Otto von Bismarck.
Today, we take our first look ever at a small developmental concern. Recent trial results have boosted the shares substantially and also drawn in some insider buying. An analysis follows below.
Company Overview
Dyne Therapeutics, Inc. (NASDAQ:DYN) is a Waltham, Massachusetts based early clinical-stage biopharmaceutical concern focused on the development of oligonucleotide (small bits of RNA or DNA molecules) therapies that treat genetic diseases of the muscle. It has two programs targeting muscular dystrophy indications that entered the clinic in 3Q22. Dyne was formed in 2017 and went public in 2020, raising net proceeds of $246.4 million at $19 a share. The stock trades near $13.50 a share, translating to a market cap of approximately $700 million.
September Company Presentation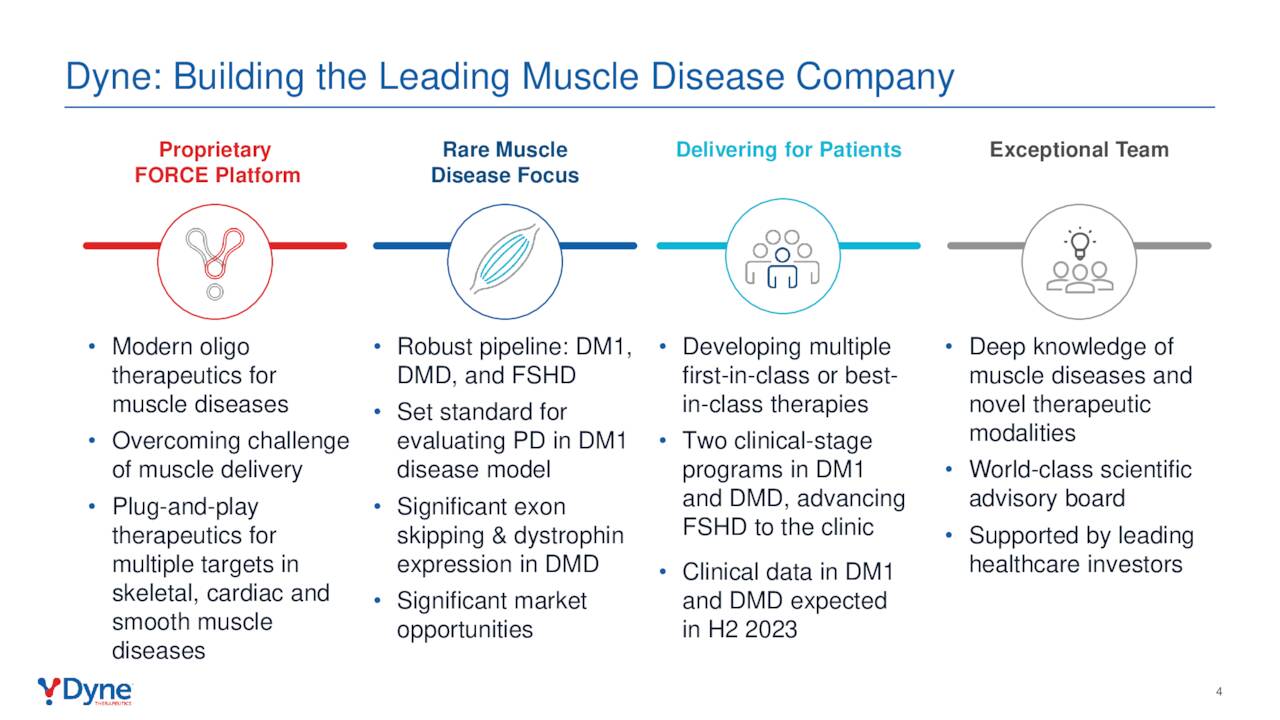
FORCE Platform
The company’s therapies are all spawned from its FORCE platform, which utilizes the muscle cells’ own Transferrin 1 receptors (TfR1) to deliver its payload. Its therapeutics consist of three components: an antigen binding fragment [FAB]; an oligonucleotide payload; and a linker that connects the two. The Fab is engineered to join to the TfR1 receptor, which is highly expressed on the surface of muscle cells. That bind acts as the entry mechanism into the cell, where a bioactive moiety of either an antisense oligonucleotide [ASO, small interfering RNA (siRNA), or phosphorodiamidate morpholino oligomer [PMO] can be delivered. This endosomal escape is accomplished without any membrane-altering agents – significant in that the endosomal uptake pathway is designed to degrade these molecules; thus reducing their bioavailability. Each therapy employs the same Fab and linker, permitting modularity across the FORCE platform.
September Company Presentation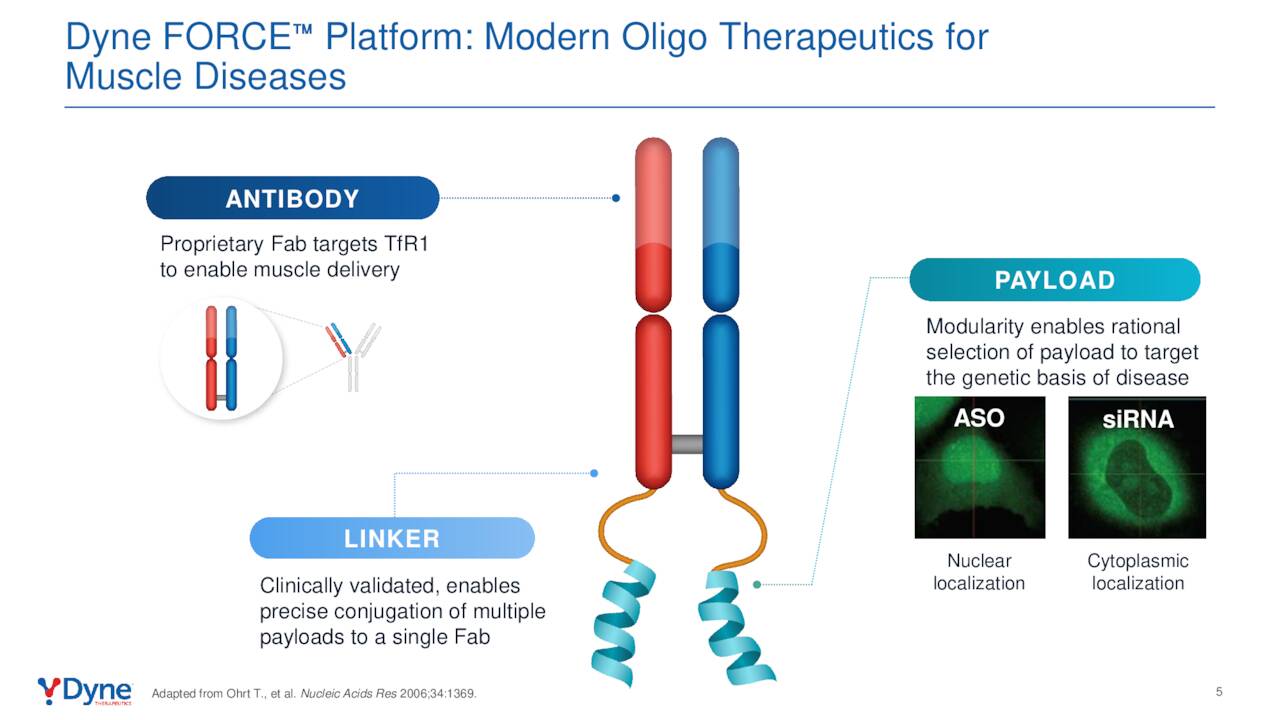
Pipeline
To date, FORCE has produced two clinical candidates.
September Company Presentation
DYNE-101. The company’s co-lead program is DYNE-101, which targets myotonic dystrophy type 1 (DM1), a monogenic, autosomal dominant disease characterized through a myriad of manifestations – including myotonia, muscle weakness, cardiac arrhythmias, fatigue, GI issues, cataracts, and cognitive impairment – resulting from damage to skeletal, cardiac, and smooth muscles. It is caused by an increase in the number of CTG triplet repeats found in the 3’ non-coding region of the DMPK gene. The number of repeats in healthy subjects is ~35 versus thousands in DM1 patients. The abnormal expansion of the gene forms hairpin loops that entrap the pre-mRNA in the nucleus, driving toxic activity known as a gain-of-function mutation. The toxic mRNA forms foci (RNA repeats) in the nucleus that bind to splicing proteins. With the splicing proteins unable to perform their role in translation, other mis-spliced proteins are created, causing the aforementioned presentations of DM1.
September Company Presentation
DM1 afflicts more than 40,000 in the U.S. and ~74,000 in Europe, with about two-thirds presenting symptoms between their early teens to 50 years of age. There are no approved therapies.
Dyne believes a reduction in DMPK levels will permit normal mRNA processing and translation of normal proteins, which could stop or reverse disease progression. DYNE-101 is designed to translocate to the nucleus and target toxic nuclear DMPK RNA, resulting in its degradation. After showing promise in animal models in the preclinic, it was entered into a Phase 1/2 study in September 2022. Dubbed ACHIEVE, it is evaluating the safety and tolerability of DYNE-101 in ~64 DM1 patients between 18 and 49 years of age. The trial is comprised of a 24-week multiple ascending dose [MAD], placebo-controlled period, a 24-week open-label extension, and a 96-week long-term extension. Designed to be potentially registrational, secondary endpoints include pharmacokinetics and change from baseline in splicing DMPK RNA expression, and assessments of muscle strength and function. Safety and splicing data are anticipated in 2H23.
DYNE-251. The company’s other co-lead asset is DYNE-251, which is designed to treat Duchenne muscular dystrophy (DMD), a monogenic, X-linked disease caused by mutations that encode for the dystrophin protein. Essential for the structural integrity and function of muscle cells involved in walking, breathing, and cardiac function, dystrophin proteins are reduced or absent in DMD patients due to mutations in the dystrophin gene. Symptoms such as muscle weakness and wasting present in the first few years of life with most patients wheelchair-bound by their teenage years. Progressing to respiratory and cardiac failure, nearly all sufferers are dead by the age of 30.
Made famous by the Jerry Lewis Labor Day Telethons, DMD afflicts ~12,000 to 15,000 in the U.S. and ~25,000 in Europe. In addition to PTC Therapeutics’ (PTCT) corticosteroid Emflaza, there are four FDA-approved exon skipping therapies for the affliction. These remedies encourage the cellular machinery to skip over missing, faulty, or misaligned sections (exons) of genetic code, leading to truncated but functional proteins. Sarepta Therapeutics has developed three ‘patches’ that promote skipping of Exons 45, 51, 53 of the dystrophin gene.
Dyne believes its Fab targeting TfR1 conjugated to a single stranded PMO will deliver said PMO more efficiently than Sarepta’s approach, meaning (potentially) more dystrophin expression, and less frequent dosing. Its first foray employing it exon skipping strategy is DYNE-251, which promotes Exon 51 skipping. Although, Sarepta’s Exondys 51 (eteplirsen) is approved for DMD patients amenable to Exon 51 skipping, it does not meaningfully elevate dystrophin production (<1%).
September Company Presentation
After demonstrating that it could overcome the delivery limitations of approved therapies by significantly raising dystrophin levels in preclinical animal models, DYNE-251 entered the clinic in September 2022, where it is undergoing evaluation in a Phase 1/2 study (DELIVER). The trial is similar to ACHIEVE – both in design and its potentially registrational impact – consisting of ~46 male DMD patients who are amenable to Exon 51 skipping aged 4 to 16. Primary efficacy endpoint is change from baseline in dystrophin levels as measured by Western blot. Secondary endpoints include muscle function measures, exon skipping, and pharmacokinetics. Dystrophin level data from the MAD placebo-controlled portion of DELIVER is expected in 2H23.
DNYE-251 received Fast Track designation from the FDA in October 2022.
September Company Presentation
It should be noted that the FDA initially put a clinical hold on Dyne’s IND application for DYNE-251, requesting more data. This development delayed its entry into the clinic by approximately six months. During the hold, which was announced on January 18, 2022, shares of DYN fell to an all-time low of $4.30 (June 2, 2022), at which point they were trading at a discount to cash. They have been on an upward trajectory since, buoyed by the FDA clearances of both DYNE-251 and DYNE-101 IND applications in July 2022.
September Company Presentation
The company has also preclinically advanced DYNE-301 for the treatment of facioscapulohumeral dystrophy, a genetic muscle disorder predominantly affecting the face, shoulder blades, and upper arms. It was initially scheduled for an IND filing in 2H22 but was deprioritized in favor of allocating resources to advance the company’s DYNE-101 and DYNE-251 programs.
Balance Sheet & Analyst Commentary:
To advance those candidates, Dyne held cash and marketable securities of $248.1 million as of September 30, 2022, providing it a runway through YE24. The company has a $150 million ATM facility at its disposal but (to date) has elected not to utilize it.
The Street is unanimously bullish on Dyne’s prospects, featuring four buy and three outperform ratings. That said, their median 12-month price objective is a somewhat uninspiring $17.50.
Current Chairman of the Board and past (founding) CEO Jason Rhodes, who also represents the interest of Atlas Venture, is bullish. On behalf of Atlas, he purchased 934,581 shares at an average price of $10.70, upping its ownership interest to 19%. The $10 million investment was well-timed, with news of positive early data from rival Avidity Biosciences’ (RNA) Phase 1/2 DM1 study on December 14th – 45% mean reduction in DMPK and 31% improvement DM1-related splicing after a single dose – sending shares of DYN upward 16% since their December 12th close.
Verdict:
The reason Avidity’s press release propelled Dyne Therapeutics, Inc. stock higher lies in the fact that they use similar Fab-linker-oligonucleotide approaches for treating DM1, with the former’s compounds called antibody oligonucleotide conjugates. Although attacking the same disease((s)), the news somewhat de-risks Dyne’s approach.
That said, it will be at least six months (maybe longer) before Dyne Therapeutics, Inc.’s data releases for DM1 and DMD, meaning a catalyst vacuum that could cause its stock to drift lower. Dyne has a potentially exciting approach in an area of high unmet need but after a larger than 200% move off its June 2022 low, there is likely to be an opportunity to buy it closer to $10 over the subsequent months. As such, we’ll keep an eye on Dyne Therapeutics, Inc. as the data releases draw nearer.
War is sweet to those who have not experienced it.”― Desiderius Erasmus


Be the first to comment