Naeblys
To be trusted is a greater compliment than being loved.― George MacDonald
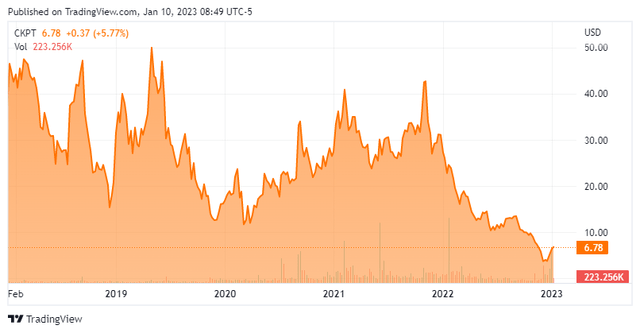
Today, Checkpoint Therapeutics, Inc. (NASDAQ:CKPT) goes back into the spotlight for the first time since our initial article on it in February of last year. There has been some significant news flow around this clinical-stage immunotherapy and targeted oncology company in recent months. An updated analysis follows below.
Company Overview
Checkpoint Therapeutics is located just outside of Boston. The company is focused on developing novel treatments for patients with solid tumor cancers. The company’s lead antibody product candidate is called Cosibelimab. According to the company’s website, Cosibelimab consists of:
A fully-human monoclonal antibody of IgG1 subtype that directly binds to programmed death ligand-1 (PD-L1) and blocks the PD-L1 interaction with the programmed death receptor-1 and B7.1 receptors.
September Company Presentation
The company is also developing CK-302. This candidate is a human agonistic antibody that is designed to bind to and trigger signaling in GITR expressing cells. It has several other earlier stage assets in development as well. The stock currently trades just under seven bucks a share and sports an approximate market capitalization of $130 million.
Recent Developments
When we last visited Checkpoint, the company had just posted positive topline results from its registration-enabling clinical trial for cosibelimab in late January. This study was evaluating the safety and efficacy of cosibelimab to treat metastatic cutaneous squamous cell carcinoma (cSCC) patients.
September Company Presentation
In June, interim data from a study showed an impressive objective response rate or ORR in 31 patients of 54.8% to treat locally advanced cSCC. Based on this, the company plans to file a Biologic License Application or BLA for both this indication along with metastatic cutaneous squamous cell carcinoma for which trial results showed a 47.4% ORR.
September Company Presentation
cSCC is a significant potential market (approximately $1 billion by 2024) and cosibelimab compares favorably to current treatments and will be marketed at a significantly lower price point (approximately 30% less in the U.S. and 20% in Europe) to other PD-1 focused treatments.
September Company Presentation
In mid-May, the European Medicines Agency and the U.K. Medicines & Healthcare products Regulatory Agency both issued Pediatric Investigation Plan or PIP product-specific waivers granted for cosibelimab in the EU and U.K. As H.C. Wainwright noted at the time ‘The granted waiver should, in our opinion, support future regulatory review (in both the U.S. and abroad), reduce development costs and expenses, and increase the overall value” of the program, especially in Europe‘
Analyst Commentary & Balance Sheet
There has been a noticeable uptick in analyst firm activity in the past three weeks. B. Riley Financial has raised its price target from $7 to $15 while maintaining their Buy rating on the stock. Both H.C. Wainwright ($76 price target) and Cantor Fitzgerald have also reissued Buy ratings. Cantor also highlighted Checkpoint as a possible takeover target in October.
Approximately five percent of the outstanding float is currently held short. Various insiders sold nearly $700,000 worth of stock in the first half of 2022. There has been no insider activity in the stock since then.
The company ended the third quarter with $20.5 million in cash and marketable securities on its balance sheet. It has no long term debt. Checkpoint has burned through $34.2 million worth of cash to fund operations/development in the first nine months of 2022. The company executed a direct offering in mid-December to raise an additional $7.5 million of funding. This came roughly ten days after the stock had a reverse ten to one split.
Verdict
We concluded our first article on Checkpoint stating at best the stock merited a very small “watch item” position. With the shares down significantly since then and the company making progress toward FDA approval for its lead asset, we are slightly more positive on the shares.
September Company Presentation
Cosibelimab is aiming at a significant niche of the overall PD-1 market. Checkpoint should file the BLA for two indications by the end of this month with a marketing application for Europe to follow. Cosibelimab has the potential to be the first anti-PD-L1 antibody approved for advanced cSCC, which is the second most frequently diagnosed skin cancer.
September Company Presentation
The company is also evaluating its lead small-molecule, a targeted anti-cancer agent called olafertinib. Checkpoint sees this as a potential new treatment for patients with EGFR mutation-positive non-small cell lung cancer as olafertinib is a third-generation epidermal growth factor receptor or EGFR inhibitor.
September Company Presentation
In short, there are some positive developments at Checkpoint. That said, based on its burn rate and upcoming milestones, the company is going to have to raise significant additional capital to get Cosibelimab approved as well as its potential initial rollout in 2024. This most likely will be substantially dilutive to current shareholders. I will probably add to my minute stake in CKPT when the next fund raising event occurs as the shares long term risk/reward profile looks potentially compelling at these levels for speculative investors.
September Company Presentation
Trusting is hard. Knowing who to trust, even harder.”― Maria V. Snyder


Be the first to comment