
ipopba
There are two kinds of courage the courage to die and the courage to live”― Sakue Shimohira
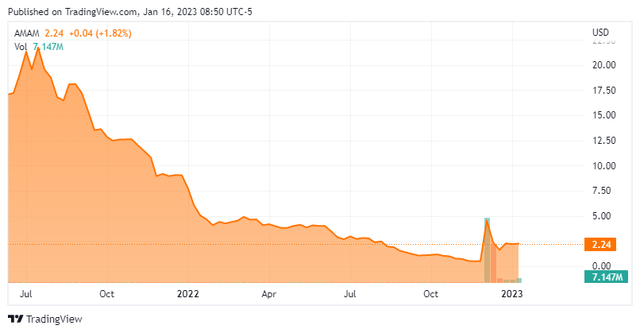
Today, we take our first look at Ambrx Biopharma Inc. (NYSE:AMAM). The stock of this oncology focused developmental firm has been under considerable pressure since the company came public in mid-2021 in the middle of an IPO wave. However, Ambrx posted some encouraging trial results this December. Is the start of a turnaround for the company’s fortunes and that of its shareholders? An analysis follows below:
Seeking Alpha
Company Overview
Ambrx Biopharma Inc. is based just outside of San Diego in La Jolla, CA. This developmental concern has several drug candidates in its pipeline targeted various indications of cancer. These pipeline assets were developed using an expanded genetic code technology platform to create Engineered Precision Biologics or EPBs.
May Company Presentation
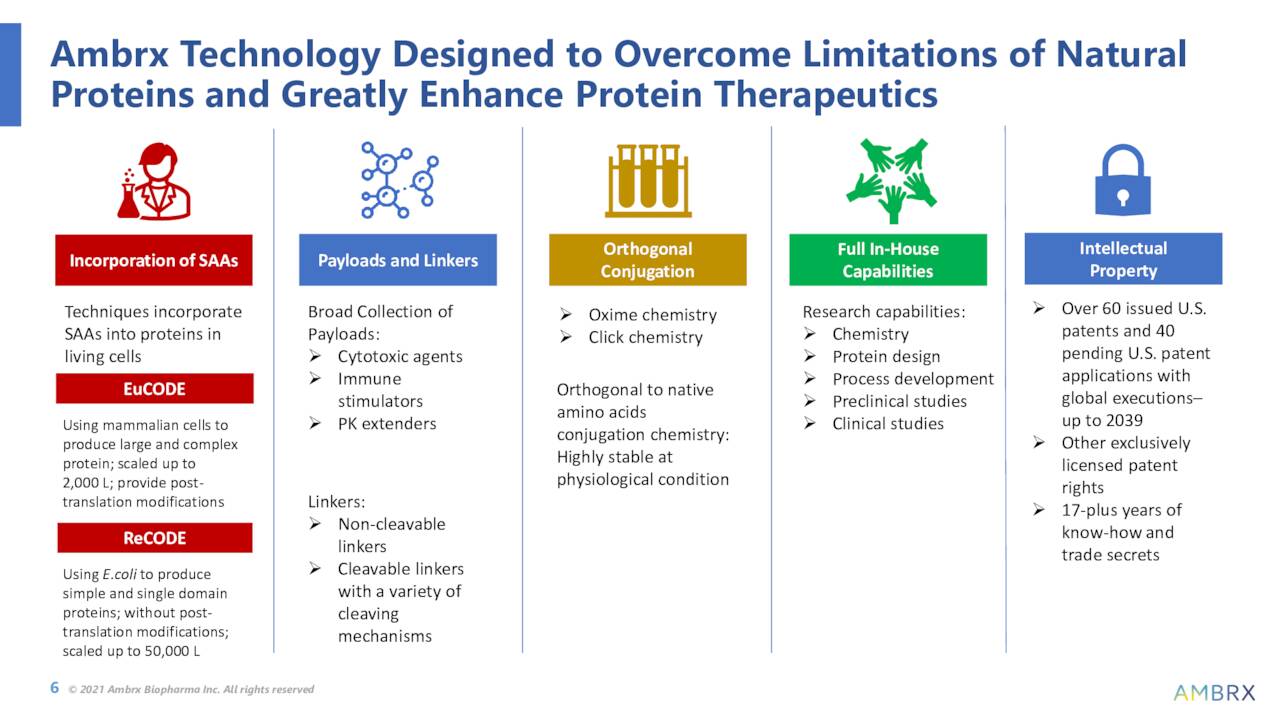
The company is working to incorporate synthetic amino acids into proteins within living cells. It is hoped that this can overcome the limitations of conventional conjugation approaches to treating different forms of cancers.
May Company Presentation
Ambrx also is partnered with larger drug makers like Bristol-Myers Squibb (BMY) in its developmental efforts. The stock trades just above two bucks a share and sports an approximate market capitalization of $85 million.
May Company Presentation
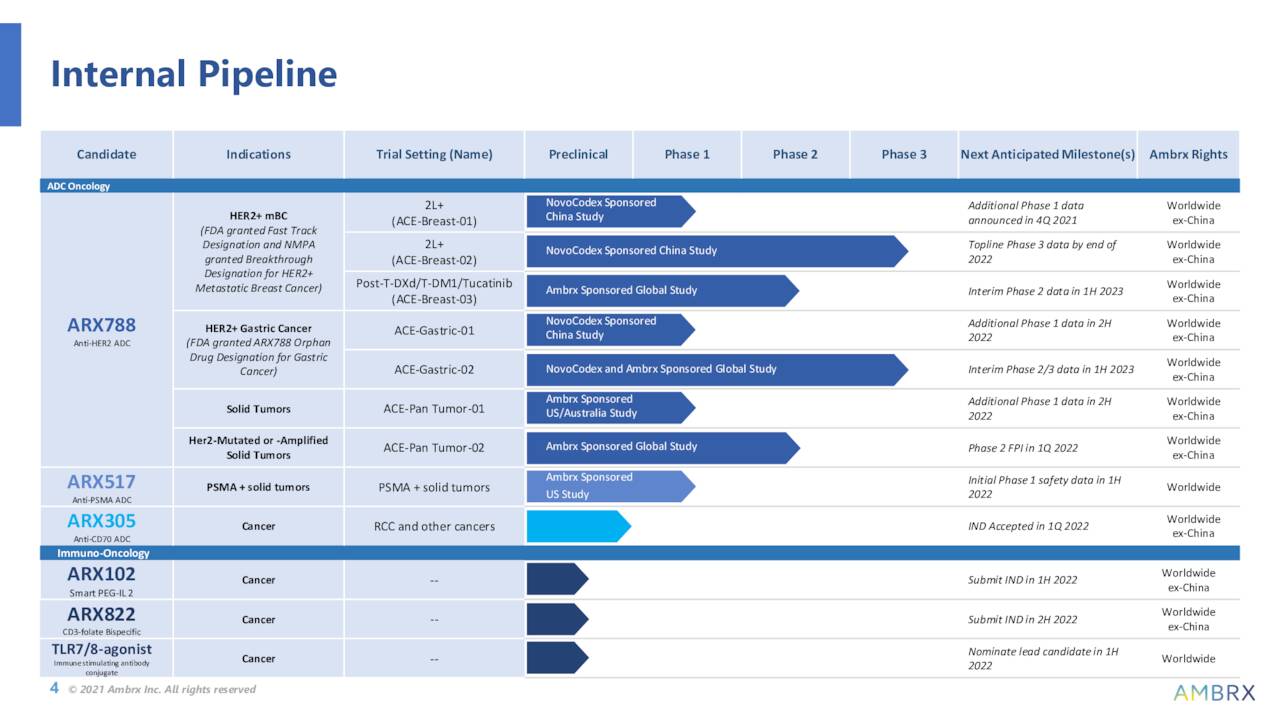
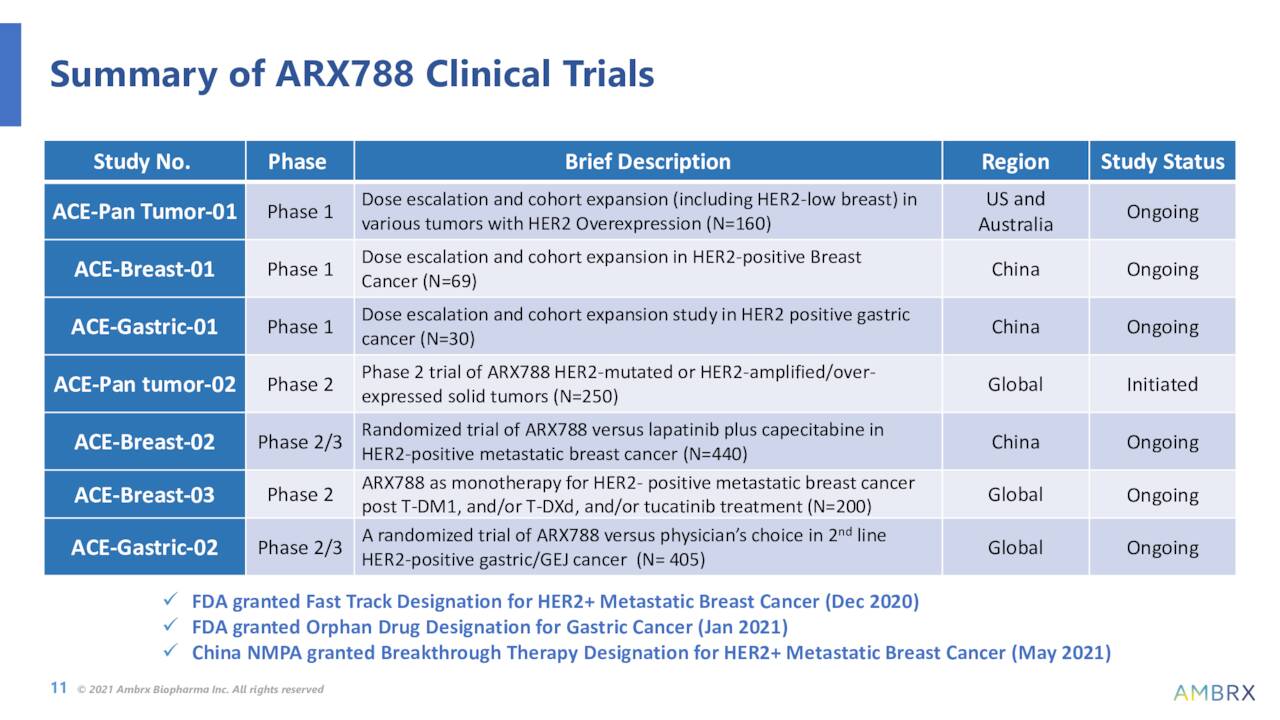
The company’s initial focus is on developing antibody drug conjugates (ADCs) to modulate the immune system that can lead to potential treatments for various cancer indications with a high unmet medical need. Ambrx’s most advance asset, until recently, is called ARX788. This candidate is an anti-HER2 ADC. It is currently being studied broadly in breast cancer, gastric/GEJ cancer and other solid tumor clinical trials in conjunction with Ambrx’s developmental partner NovoCodex Biopharmaceuticals in China. Positive Phase 1 data targeting breast cancer was posted late in 2021.

May Company Presentation
According to the company’s website ARX788:
Is a homogeneous and highly stable ADC, which targets the HER2 receptor and contains two AS269 cytotoxic payloads site-specifically conjugated to a trastuzumab-based antibody. ARX788 was designed to maximize potential anti-tumor activity by optimizing the number and position of the payloads and the chemical bonds that conjugate the payloads to the antibody.”
May Company Presentation
Notably, ARX788 has Fast Track Designation in HER2+ metastatic breast cancer and Orphan Drug Designation in gastric cancer.
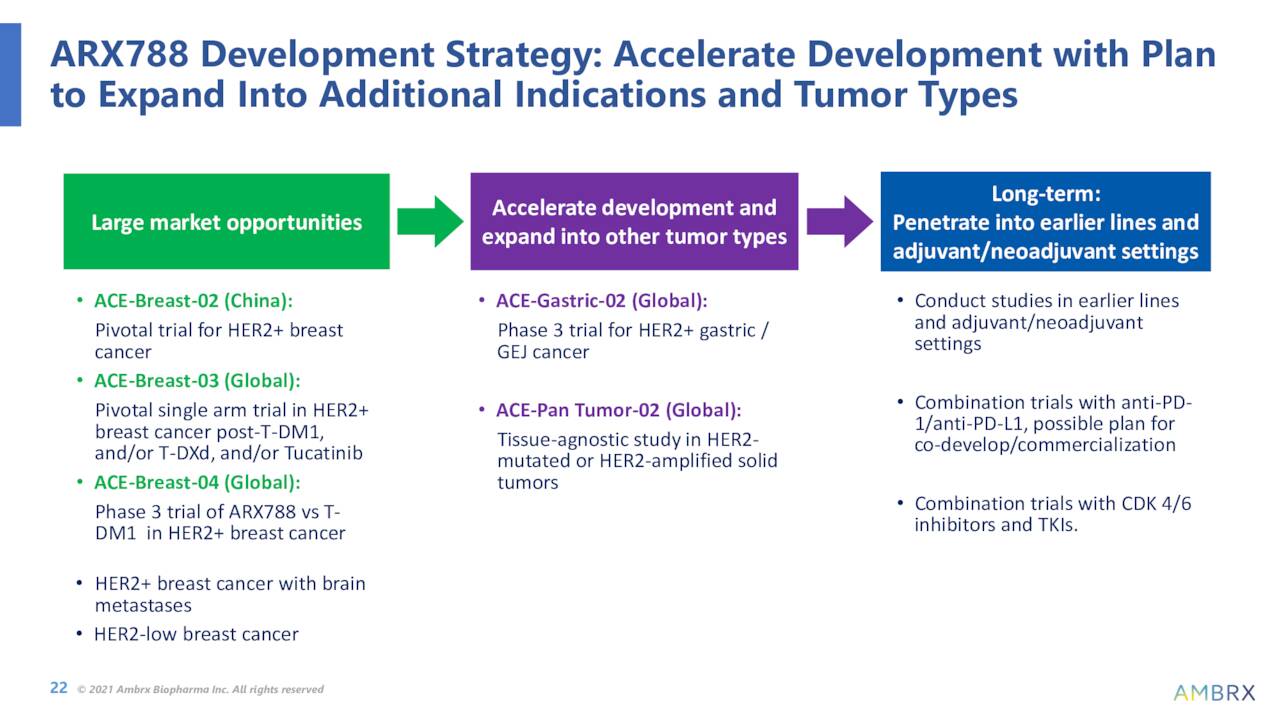
The company’s lead wholly owned asset is named ARX517 and this compound was created to target the prostate-specific membrane antigen (PSMA) expressed on prostate cancer cells. Interim safety data from a Phase 1 study should be sometime in the second half of this year. Another compound (ARX305) recently got IND clearance from the FDA and Ambrx plans to advance it to a Phase 1 trial sometime this year. The company has several other assets in very stage development.
Recent Developments:
2022 was certainly an eventful year for Ambrx Biopharma Inc. and its shareholders. The company named a new Chief Medical Officer in May. It then replaced its CEO, who had been with the company since 2002 with a new interim leader in August which was followed by a new CEO in early November. In the midst of the CEO transition, the company announced a major restructuring in October.
The company will cut 15% of its workforce to reduce costs. More importantly, Ambrx will halt internal development of ARX788 until it finds a partner outside of China. Earlier stage ARX517 will be ‘promoted‘ to be the company’s new ‘lead asset‘. These actions are intended to extend Ambrx’s cash runway into 2025.
It should be noted, the company at least ended 2022 on a high note. On December 9th, the company announced that a Phase 2 trial around ARX788 to treat HER2+ metastatic breast cancer who were previously treated with Roche’s (OTCQX:RHHBY) Kadcyla had positive results with no severe adverse effects. Patients treated with ARX788 had a confirmed ORR of 57.1% and unconfirmed ORR of 71.4%. This news triggered the large rise in the shares over the past five weeks, even if the trial only involved seven patients.
Analyst Commentary & Balance Sheet
Only three analyst firms have chimed in around Ambrx Biopharma over the past year. Goldman Sachs last maintained its Hold rating and $4 price target on AMAM on May 24th. Roughly four months later, Robert W. Baird reissued its Buy rating on the stock but cut its price target in half to four bucks a share. Finally, on December 11th, H.C. Wainwright reiterated its own Buy rating on the shares.
Approximately 15% of the stock’s outstanding float is currently held short. The company last reported quarterly results when it posted second quarter data in late August. At that time, the firm had nearly $130 million on its balance sheet worth of cash and marketable securities. Ambrx burned through nearly $50 million to fund operations in the first six months of 2022. The company posted a net loss of $51.5 million for the first two quarters of the year while posting $2.9 million worth of revenue from R&D and license agreements.
Verdict
It is notable that Goldman Sachs has a Hold rating on the firm given the investment bank was the lead underwriter of the company’s IPO back in June of 2021.
May Company Presentation
That said, the stock trades for less than the cash/marketable securities on its balance sheet and has a cash runway into 2025. Several data readouts from the trials its partner NovoCodex Biopharmaceuticals is running in China should be out sometime in 2023. These consist of two phase 3 studies and one phase 2 trial of ARX788 in breast cancer and gastric/GEJ cancers.
The recent management turnover and major company restructuring hardly provides much confidence for investors even given Ambrx’s pipeline potential promise. Until Ambrx finds a domestic partner for development of ARX788, the shares can only be considered for investment by aggressive, speculative investors in my opinion.
Stubbornness is determination stripped of wisdom.”― Craig D. Lounsbrough
Editor’s Note: This article discusses one or more securities that do not trade on a major U.S. exchange. Please be aware of the risks associated with these stocks.


Be the first to comment